| Line 14: | Line 14: | ||
<div class="igem_2018_team_content" style="background-color: white"> | <div class="igem_2018_team_content" style="background-color: white"> | ||
| − | + | ||
| − | + | ||
<!-- BEGIN CONTENT ---------------------------------------------------> | <!-- BEGIN CONTENT ---------------------------------------------------> | ||
<img src="https://static.igem.org/mediawiki/2018/1/16/T--Groningen--banner_results.png" class="responsive-img"> | <img src="https://static.igem.org/mediawiki/2018/1/16/T--Groningen--banner_results.png" class="responsive-img"> | ||
<br></br> | <br></br> | ||
| + | <div class="igem_2018_team_column_wrapper"> | ||
| + | <div class="column"> | ||
<div class="boxed"> | <div class="boxed"> | ||
<h4>Achievements</h4> | <h4>Achievements</h4> | ||
<h6><font size = "4.4"><b> - Enzymatic degradation of cellulose, phosphorylated cellulose and Recell. | <h6><font size = "4.4"><b> - Enzymatic degradation of cellulose, phosphorylated cellulose and Recell. | ||
| − | + | <br> - Growth of <i>Saccharomyces cerevisiae</i> on 1-4 beta linked glucose in the form of cellobiose. | |
| − | + | <br> - Conversion of phenylalanine to trans-cinnamate using PAL2. | |
| − | + | <br> - Styrene production from glucose using <i>S. cerevisiae</i>.</b</font></h6> | |
</div> | </div> | ||
<h4>Our EGII can degrade cellulose and pretreatments are effective</h4> | <h4>Our EGII can degrade cellulose and pretreatments are effective</h4> | ||
| − | <p>As raw cellulose sources such as ReCell do not have high solubility in aqueous solutions, the | + | <p>As raw cellulose sources such as ReCell do not have high solubility in aqueous solutions, the ꞵ-1,4 bonds are difficult to access for our enzymes. Therefore, we investigated different methods of cellulose pretreatment [1]. We compared enzymatic degradation of pure ReCell, ball-milled ReCell, pure cellulose and phosphorylated pure cellulose. On these cellulose sources, we tested three purchased enzyme isozymes of the cellulases used in our design: CbhI from <i>Trichoderma sp.</i>, EgII from <i>Aspergillus niger</i> (EgA) and BglI from <i>A. niger</i>. As a positive control, we used a purchased cellulose enzyme blend containing a mixture of cellulases. In a follow-up assay we tested our own purified EgII from <i>Trichoderma reesei</i> (EgII), which was expressed in <i>E. coli</i> and isolated using His-tag purification.</p> |
| − | <p> | + | <figure><img src="https://static.igem.org/mediawiki/parts/0/02/T--Groningen--egii_wb.png"><figcaption><i>Figure 1. CBP-Star Western blot of His-EgII purification fractions. The following samples were used for analysis: insoluble fraction of the cell lysate (P), soluble fraction of the cell lysate (SF), soluble fraction after incubation with Nickel beads (nSF), flow through after filtration through a disposable column (FT), two wash fractions (W1, W2) and the two elution fractions (EF1, EF2).</i></figcaption></figure> |
| − | <p>In the first experiment, we incubated the three purchased cellulases separately and in a mix (representing the cellulosome mixture) with the different cellulose sources; in this manner we investigate which pretreatments would enhance the enzymatic degradation. Overnight incubation of the cellulases and cellulose resulted in the chromogenic pattern as can be seen below. The positive control contains such large amounts of cellulases that it becomes pink under all conditions tested (it is brownish because the blend has a brown color). Out of the types of cellulose tested phosphorylated cellulose shows the most degradation, especially by | + | <p>There is a considerable amount of our protein present in the insoluble fraction, but most is present in the soluble fraction from which it can be purified. The bands at 44kDa for the elution fractions correspond with the expected size of His-EgII. Therefore, we conclude we have successfully expressed and purified His-EgII from our <i>E. coli</i> strain.</p> |
| − | <figure><img src="https://static.igem.org/mediawiki/2018/4/4b/T--Groningen--cellulase_assay.png | + | <p>A colorimetric cellulose hydrolysis assay was used to measure cellulose degradation. The assay uses a modified Chito-oligosaccharide oxidase that specifically oxidizes the C1 hydroxyl moiety of cellulose hydrolytic products. The oxidation produces H2O2 in equimolar amounts which is used by horseradish peroxidase to produce a color that is stoichiometrically equivalent to the amount of hydrolytic events [2].(read more about the assay <a href="https://2018.igem.org/Team:Groningen/Protocols#galactose-induction" target="_blank">here</a>.</p> |
| − | <p> | + | <p>In the first experiment, we incubated the three purchased cellulases separately and in a mix (representing the cellulosome mixture) with the different cellulose sources; in this manner we investigate which pretreatments would enhance the enzymatic degradation. Overnight incubation of the cellulases and cellulose resulted in the chromogenic pattern as can be seen below. The positive control contains such large amounts of cellulases that it becomes pink under all conditions tested (it is brownish because the blend has a brown color). Out of the types of cellulose tested phosphorylated cellulose shows the most degradation, especially by CbhI. Pure cellulose and ball-milled ReCell can be degraded as well, but this requires more time than degrading phosphorylated pure cellulose. Unprocessed ReCell is not visibly degraded under the conditions tested here. The mix of the three enzymes was just as effective as CbhI, but degradation of this mix is likely mostly due to CbhI, since EgA and BglI did not show visible activity alone under these conditions.</p> |
| − | <p>In a following assay we tested the endoglucanases specifically. We compared our own purified | + | <figure><img src="https://static.igem.org/mediawiki/2018/4/4b/T--Groningen--cellulase_assay.png"><figcaption><i>Figure 2. Row A to D contain pure ReCell, ball-milled ReCell, pure cellulose and phosphorylated cellulose, respectively. Lanes 1 to 6 contain BglI, EgA, CbhI, a mix of 1-3, the positive control and negative control, respectively.</i></figcaption></figure> |
| − | <figure><img src="https://static.igem.org/mediawiki/2018/f/fe/T--Groningen--cellulase_assay_EGII.png | + | <p>EgA, CbhI and BglI were added in the same concentrations, but CbhI showed much higher enzyme activity.To obtain synergistic activity levels, the expression ratio of the cellulosome enzymes can be fine tuned, for example by use of different promoters. Follow-up experiments should determine an optimal ratio of the three enzymes, perhaps while bound to a recombinantly expressed scaffold molecule. Phosphorylated cellulose was degraded more efficiently than ball-milled ReCell, however, the process does require chemicals whereas ball milling does not. Therefore, our <a href="https://2018.igem.org/Team:Groningen/Design" target="_blank">bioreactor design</a> applies ball-milling instead of phosphorylation.</p> |
| − | <p>Again, we see degradation by the positive control on all substrates. However, the purchased | + | <p>In a following assay we tested the endoglucanases specifically. We compared our own purified EgII with the purchased EgA, on the same cellulose substrates used in the assay above. We tested undiluted EgII, EgA in approximately the same concentration, and 1:1000 EgII and 1:10.000 diluted EgII to estimate the enzyme activity. </p> |
| − | + | <figure><img src="https://static.igem.org/mediawiki/2018/f/fe/T--Groningen--cellulase_assay_EGII.png"><figcaption><i>Figure 3. Row A to D contain pure ReCell, ball-milled ReCell, pure cellulose and phosphorylated cellulose, respectively. Lanes 1 to 6 contain EgA, EgII, EgII 1:1000, EGII 1:10.000, positive control and negative control, respectively. </i></figcaption></figure> | |
| − | <h4><i>S. cerevisiae</i> strain containing artificial cellulosome can grow on cellobiose</h4> | + | <p>Again, we see degradation by the positive control on all substrates. However, the purchased EgA enzyme did not show much activity. Our undiluted purified EgII did show activity on all substrates except for unprocessed ReCell. This is surprising, because we expected that the purchased EgA contains more enzyme units than our EgII and as such should have a higher activity. The results show that our own cellulase has cellulolytic activity and highlight the fact that preprocessing of cellulose or ReCell for enzymatic processes is highly beneficial for the degradation process.</p> |
| + | <h4><i>S. cerevisiae</i> strain containing artificial cellulosome can grow on cellobiose and phosphorylated cellulose</h4> | ||
<p>In order to demonstrate that our strain <i>S. cerevisiae BJ1991</i> expressing the cellulosome (BJ1991+) is able to degrade cellulose and grow, a growth assay is performed. Because long strands of cellulose are poorly soluble, a growth assay is performed on cellobiose. Cellobiose is a glucose dimer, in essence the shortest glucose polymer and form of cellulose. Cells are grown on fructose induced with galactose for 6 hours, after which the cells are pelleted, washed and transferred to medium containing cellobiose. The strains are loaded in a plate reader with different starting OD600s, and measured over a 24 hour timespan.</p> | <p>In order to demonstrate that our strain <i>S. cerevisiae BJ1991</i> expressing the cellulosome (BJ1991+) is able to degrade cellulose and grow, a growth assay is performed. Because long strands of cellulose are poorly soluble, a growth assay is performed on cellobiose. Cellobiose is a glucose dimer, in essence the shortest glucose polymer and form of cellulose. Cells are grown on fructose induced with galactose for 6 hours, after which the cells are pelleted, washed and transferred to medium containing cellobiose. The strains are loaded in a plate reader with different starting OD600s, and measured over a 24 hour timespan.</p> | ||
| − | <figure><img src="https://static.igem.org/mediawiki/2018/7/75/T--Groningen--Cellobiose_assay_fig1-2.png | + | <figure><img src="https://static.igem.org/mediawiki/2018/7/75/T--Groningen--Cellobiose_assay_fig1-2.png"><figcaption><i>Figure 4. Growth curves of BJ1991 strains containing the artificial cellulosome, after 6 hours of galactose induction, along with controls. Numbers in legend indicate starting OD600.</i></figcaption></figure> |
| − | <p>As shown in figure | + | <p>As shown in figure 4, the BJ1991+ strains are able to grow on cellobiose after 6 hours of galactose induction. Strains with a higher starting OD600 grow faster, and reach a higher OD600 before plateauing. The positive control, containing an additional 0.5% glucose, shows the most growth, and the negative control (wild-type strain) shows no growth. The induced strain without cellobiose also does not show any growth, indicating that residual galactose is not responsible for growth. The uninduced strain with cellobiose does show a little bit of growth, although much less than the induced strains. This is likely caused by leakage of the promoter.</p> |
| − | <figure><img src="https://static.igem.org/mediawiki/2018/7/74/T--Groningen--Cellobiose_assay_fig2-2.png | + | <figure><img src="https://static.igem.org/mediawiki/2018/7/74/T--Groningen--Cellobiose_assay_fig2-2.png"><figcaption><i>Figure 5. Growth curves of BJ1991 strains containing the artificial cellulosome, with start OD600 of 0.1, with different galactose induction durations.</i></figcaption></figure> |
| − | <p>We also compared growth with different galactose induction times. As shown in figure | + | <p>We also compared growth with different galactose induction times. As shown in figure 5, samples with equal starting OD600 show faster growth when the galactose induction time is longer. The strain induced for 6 hours grows approximately 2 times faster than the strain induced for 2 hours. To obtain even higher growth rates longer induction can be used, as induction for longer than 6 hours has not been tested.</p> |
| − | < | + | <p>We attempted growing our strain on cellulose and phosphorylated cellulose, but were unable to detect growth on pure cellulose and phosphorylated cellulose in a plate reader. Absence of growth on pure cellulose is expected as cellulose is notoriously difficult to degrade [1]. However, growth using the cellulosome on phosphorylated cellulose has been shown before [3]. We hypothesized there was no growth due to poor stirring in the plate reader and grew our strain in a flask. Now we were able to grow our strain on phosphorylated cellulose in a flask. A negative control the same strain without carbon source was grown.</p> |
| + | <p><i>Table 1. Growth on phosphorylated cellulose using YPH499 containing PAL2 and the cellulosome (YPH499++). OD600 was measured at t=0, t=12h and t=36h. Initial OD600 value is high due to absorbance from the phosphorylated cellulose.</i> </p> | ||
| + | <table><tr><th></th><th>OD600 at t=0</th><th>OD600 at t=12h</th><th>OD600 at t=36h</th></tr><tr><td>YPHYPH499++</td><td>0,51</td><td>0,55</td><td>1,36</td></tr><tr><td>negative control</td><td>0,1</td><td>0,06</td><td>0,1</td></tr></table> | ||
<h4>Styrene Detection with RP-HPLC UV DAD</h4> | <h4>Styrene Detection with RP-HPLC UV DAD</h4> | ||
| − | <p>After testing <a href=" | + | <p>After testing <a href="http://parts.igem.org/wiki/index.php?title=Part:BBa_K2783010" target="_blank"><i>PAL2</i> in <i>E. coli</i></a>, we tested styrene production in <i>YPH499</i> with <i>PAL2</i> using RP-HPLC UV DAD; we based our method on an established protocol [4]. This HPLC separates compounds based on their affinity for the column and measures their absorbance spectrum. A test run gave us the elution conditions and retention time of styrene. Comparing the UV spectrum of the fraction with the known UV spectrum of styrene proved the fraction was indeed styrene.</p> |
| − | <figure><img | + | <figure><img src="https://static.igem.org/mediawiki/2018/6/68/T--Groningen--Styrene_spectra.png"><figcaption><i>Figure 6. UV spectrum of the peak at 17.5 min retention time. The styrene absorption spectrum from literature is added in the top right [5].</i></figcaption></figure> |
| − | <p>As can be seen in | + | <p>This resulted in the following HPLC method for styrene detection: using a gradient of methanol and ddH2O with 30% methanol at the start to 100% methanol over 45 minutes. The retention time of styrene under these conditions was determined to be 17,5 min. The strains were grown for two days on 2% glucose and styrene production was boosted with 0,5 g/L phenylalanine (L-phenylalanine). Four different cultures were prepared: a control with phenylalanine (Ctrl), a strain containing <i>PAL2</i> with phenylalanine (PAL2+), a strain containing <i>PAL2</i> without phenylalanine (PAL2-) and a strain containing both <i>PAL2</i> and the cellulosome with phenylalanine (PAL2++). After incubation, the cell culture was centrifuged and the supernatant was loaded onto the column. Samples were also prepared using ethyl acetate extraction, but HPLC runs did not show clear peaks. As such culture supernatant was used for styrene detection. </p> |
| − | <figure><img | + | <figure><img src="https://static.igem.org/mediawiki/2018/9/9e/T--Groningen--HPLC_Graph_17_18_wholerange.png"><figcaption><i>Figure 7. HPLC 254 nm intensity plotted against time. Peaks at 10 min and 17,5 min retention time can be seen responding to trans-cinnamate and styrene respectively.</i></figcaption></figure> |
| − | <p>When we zoom in on the styrene peak we can see differences between the tested strains (Figure | + | <p>As can be seen in figure 7 a peak is observed at the retention time of 17,5 min, indicating production of styrene by our cells. Another peak can be observed at 10 minutes retention time. As this peak is not present in the control sample, we hypothesize this is trans-cinnamate. To test this we ran trans-cinnamate on the HPLC system, which indeed showed a peak at the same retention time. The trans-cinnamate peak is considerably larger than the styrene peak. We hypothesize this data indicates that the conversion from trans-cinnamate to styrene by Fdc1 is limiting under these conditions. </p> |
| − | < | + | <figure><img src="https://static.igem.org/mediawiki/2018/a/a2/T--Groningen--HPLC_Graph_17_18_min.png"><figcaption><i>Figure 8. Styrene content in culture supernatant of S. cerevisiae. The red line shows the control sample, other lines represent S. cerevisiae with PAL2 under different conditions. S. cerevisiae with PAL2 samples show a clear peak as opposed to the control, proving styrene production.</i></figcaption></figure> |
| − | <table><tr><th></th><th>OD600</th><th>Peak area (arbitrary units)</th><th>styrene/Dryweight cells (ug/mg)</th></tr><tr><td>PAL2</td><td>4.2</td><td>17887</td><td>3.773894298</td></tr><tr><td>PAL2+</td><td>2.41</td><td>35929</td><td>13.21081475</td></tr><tr><td>PAL2++</td><td>0.38</td><td>23768</td><td>55.4256197</td></tr></table> | + | <p>When we zoom in on the styrene peak we can see differences between the tested strains (Figure 8). There is a clear difference between all three <i>PAL2</i> containing strains compared to the control, indicating successful production of styrene. Both our consolidated bioprocessing strain and our <i>PAL2</i> containing strain are able to produce styrene. Comparing <i>PAL2</i> and <i>PAL2+</i> shows that addition of phenylalanine is not necessary for styrene production; however, it does boosts styrene production indicating that phenylalanine production might be a bottleneck for styrene production. In the table below, an approximation of the produced quantity of styrene can be seen. These results show we have successfully produced styrene in yeast from glucose. </p> |
| + | <i>Table 2. Approximation of styrene production per dry weight cells. Method of calculation can be seen here (link to labjournal). Peak area is corrected using a negative control. The OD600 of PAL++ was lower than expected for unknown reasons, which gives an inflated image of the amount of styrene produced by this strain.</i> | ||
| + | <table><tr><th></th><th>OD600</th><th>Peak area (arbitrary units)</th><th>styrene/Dryweight cells (ug/mg)</th></tr><tr><td>PAL2<br></td><td>4.2</td><td>17887</td><td>3.773894298</td></tr><tr><td>PAL2+</td><td>2.41</td><td>35929</td><td>13.21081475</td></tr><tr><td>PAL2++</td><td>0.38</td><td>23768</td><td>55.4256197</td></tr></table> | ||
| + | <h4>Trans-cinnamate and Styrene production from cellobiose</h4> | ||
| + | <p>As proof of concept we used our strain containing PAL2 and the cellulosome (YPH499++) to produce styrene. Cells are grown on fructose and induced with galactose for 6 hours, after which the cells are pelleted, washed and transferred to medium containing cellobiose. The same strain grown under the same conditions without cellobiose is taken as a negative control. After growth on cellobiose for 36 hours, styrene production is measured using the RP-HPLC UV DAD as described above. As can be seen from figure 9 small peaks at the retention time of trans-cinnamate and styrene, 10.3 and 17.2 minutes respectively can be observed.</p> | ||
| + | <figure><img src="https://2018.igem.org/File:T--Groningen--HPLC_Graph_cellobiose_whole.png"><figcaption><i>Figure 9. Trans-cinnamate and styrene production from cellobiose in yeast. Intensity at 254 nm is plotted against the retention time. Comparing retention time of trans-cinnamate and styrene peaks to earlier HPLC the spectrum appears to have shifted 0.3 minutes.</i></figcaption></figure> | ||
| + | <p>Zooming in on the peak at 10.2 minutes shows more trans-cinnamate is produced when growing on cellobiose in comparison to the negative control (Figure 9). A peak at the retention time of trans-cinnamate was not expected; however, as trans-cinnamate absorption is very strong at 254nm, it is possible contamination of previous samples occurred in the HPLC [6]. </p> | ||
| + | <figure><img src="https://2018.igem.org/File:T--Groningen--HPLC_Graph_transcinnamate_cellobiose.png"><figcaption><i>Figure 10. Trans-cinnamate production from cellobiose in yeast. HPLC intensity at 254 nm is plotted against the retention time. A slightly higher peak can be observed growing on cellobiose opposed to a control.</i></figcaption></figure> | ||
| + | <p>When taking a closer look at the peak corresponding to the retention time of styrene, a clear difference can be seen with the control (Figure 11). Although the peak is very small, it is higher than the background of the control. It should be noted that the supernatant of the cell cultured was used for detection of styrene. As styrene has a high affinity for apolar phases, the amount of styrene produced is much larger than can be seen on the HPLC. If time would have permitted it, we would have liked to express the cellulosome constitutively; consistent cellulosome expression should facilitate cellobiose or cellulose degradation and thus increase styrene production.</p> | ||
| + | <figure><img src="https://2018.igem.org/File:T--Groningen--HPLC_Graph_styrene_cellobiose.png"><figcaption><i>Figure 11. Styrene production from cellobiose in yeast. HPLC intensity at 254 nm is plotted against the retention time. A peak can be observed for yeast grown on cellobiose, the control spectrum is flat.</i></figcaption></figure> | ||
| + | |||
<!--References --> | <!--References --> | ||
<h4>References</h4> | <h4>References</h4> | ||
| + | <p>[1] Zhang, Y. P., Cui, J., Lynd, L. R., & Kuang, L. R. (2006). A Transition from Cellulose Swelling to Cellulose Dissolution byo-Phosphoric Acid: Evidence from Enzymatic Hydrolysis and Supramolecular Structure. Biomacromolecules, 7(2), 644-648. doi:10.1021/bm050799c </p> | ||
| + | <p>[2] Ferrari, A. R., Gaber, Y. & Fraaije, M. W. A fast, sensitive and easy colorimetric assay for chitinase and cellulase activity detection. Biotechnol. Biofuels 7, 37 (2014). </p> | ||
| + | <p>[3] Wen, F., Sun, J., & Zhao, H. (2010). Yeast surface display of trifunctional minicellulosomes for simultaneous saccharification and fermentation of cellulose to ethanol. Applied and Environmental Microbiology, 76(4), 1251–60. http://doi.org/10.1128/AEM.01687-09 </p> | ||
| + | <p>[4] Mckenna, R., Thompson, B., Pugh, S., & Nielsen, D. R. (2014). Rational and combinatorial approaches to engineering styrene production by Saccharomyces cerevisiae. Microbial Cell Factories, 13(1). doi:10.1186/s12934-014-0123-2</p> | ||
| + | <p>[5] Lei Chen, Zhiwei Xu, Jialu Li, Baoming Zhou, Mingjing Shan, Yinglin Li, Liangsen Liu, Baodong Li and Jiarong Niu: Modifying graphite oxide nanostructures in various media by high-energy irradiation. RSC Adv., 2014, 4, 1025</p> | ||
| + | <p>[6] NIST Chemistry WebBook, SRD 69. (n.d.). Retrieved from https://webbook.nist.gov/cgi/cbook.cgi?ID=C140103&Mask=400#UV-Vis-Spec</p> | ||
| + | |||
<!-- div class="line_divider"></div --> | <!-- div class="line_divider"></div --> | ||
</div> | </div> | ||
Revision as of 21:17, 17 October 2018

Achievements
- Enzymatic degradation of cellulose, phosphorylated cellulose and Recell.
- Growth of Saccharomyces cerevisiae on 1-4 beta linked glucose in the form of cellobiose.
- Conversion of phenylalanine to trans-cinnamate using PAL2.
- Styrene production from glucose using S. cerevisiae.
Our EGII can degrade cellulose and pretreatments are effective
As raw cellulose sources such as ReCell do not have high solubility in aqueous solutions, the ꞵ-1,4 bonds are difficult to access for our enzymes. Therefore, we investigated different methods of cellulose pretreatment [1]. We compared enzymatic degradation of pure ReCell, ball-milled ReCell, pure cellulose and phosphorylated pure cellulose. On these cellulose sources, we tested three purchased enzyme isozymes of the cellulases used in our design: CbhI from Trichoderma sp., EgII from Aspergillus niger (EgA) and BglI from A. niger. As a positive control, we used a purchased cellulose enzyme blend containing a mixture of cellulases. In a follow-up assay we tested our own purified EgII from Trichoderma reesei (EgII), which was expressed in E. coli and isolated using His-tag purification.

There is a considerable amount of our protein present in the insoluble fraction, but most is present in the soluble fraction from which it can be purified. The bands at 44kDa for the elution fractions correspond with the expected size of His-EgII. Therefore, we conclude we have successfully expressed and purified His-EgII from our E. coli strain.
A colorimetric cellulose hydrolysis assay was used to measure cellulose degradation. The assay uses a modified Chito-oligosaccharide oxidase that specifically oxidizes the C1 hydroxyl moiety of cellulose hydrolytic products. The oxidation produces H2O2 in equimolar amounts which is used by horseradish peroxidase to produce a color that is stoichiometrically equivalent to the amount of hydrolytic events [2].(read more about the assay here.
In the first experiment, we incubated the three purchased cellulases separately and in a mix (representing the cellulosome mixture) with the different cellulose sources; in this manner we investigate which pretreatments would enhance the enzymatic degradation. Overnight incubation of the cellulases and cellulose resulted in the chromogenic pattern as can be seen below. The positive control contains such large amounts of cellulases that it becomes pink under all conditions tested (it is brownish because the blend has a brown color). Out of the types of cellulose tested phosphorylated cellulose shows the most degradation, especially by CbhI. Pure cellulose and ball-milled ReCell can be degraded as well, but this requires more time than degrading phosphorylated pure cellulose. Unprocessed ReCell is not visibly degraded under the conditions tested here. The mix of the three enzymes was just as effective as CbhI, but degradation of this mix is likely mostly due to CbhI, since EgA and BglI did not show visible activity alone under these conditions.

EgA, CbhI and BglI were added in the same concentrations, but CbhI showed much higher enzyme activity.To obtain synergistic activity levels, the expression ratio of the cellulosome enzymes can be fine tuned, for example by use of different promoters. Follow-up experiments should determine an optimal ratio of the three enzymes, perhaps while bound to a recombinantly expressed scaffold molecule. Phosphorylated cellulose was degraded more efficiently than ball-milled ReCell, however, the process does require chemicals whereas ball milling does not. Therefore, our bioreactor design applies ball-milling instead of phosphorylation.
In a following assay we tested the endoglucanases specifically. We compared our own purified EgII with the purchased EgA, on the same cellulose substrates used in the assay above. We tested undiluted EgII, EgA in approximately the same concentration, and 1:1000 EgII and 1:10.000 diluted EgII to estimate the enzyme activity.

Again, we see degradation by the positive control on all substrates. However, the purchased EgA enzyme did not show much activity. Our undiluted purified EgII did show activity on all substrates except for unprocessed ReCell. This is surprising, because we expected that the purchased EgA contains more enzyme units than our EgII and as such should have a higher activity. The results show that our own cellulase has cellulolytic activity and highlight the fact that preprocessing of cellulose or ReCell for enzymatic processes is highly beneficial for the degradation process.
S. cerevisiae strain containing artificial cellulosome can grow on cellobiose and phosphorylated cellulose
In order to demonstrate that our strain S. cerevisiae BJ1991 expressing the cellulosome (BJ1991+) is able to degrade cellulose and grow, a growth assay is performed. Because long strands of cellulose are poorly soluble, a growth assay is performed on cellobiose. Cellobiose is a glucose dimer, in essence the shortest glucose polymer and form of cellulose. Cells are grown on fructose induced with galactose for 6 hours, after which the cells are pelleted, washed and transferred to medium containing cellobiose. The strains are loaded in a plate reader with different starting OD600s, and measured over a 24 hour timespan.

As shown in figure 4, the BJ1991+ strains are able to grow on cellobiose after 6 hours of galactose induction. Strains with a higher starting OD600 grow faster, and reach a higher OD600 before plateauing. The positive control, containing an additional 0.5% glucose, shows the most growth, and the negative control (wild-type strain) shows no growth. The induced strain without cellobiose also does not show any growth, indicating that residual galactose is not responsible for growth. The uninduced strain with cellobiose does show a little bit of growth, although much less than the induced strains. This is likely caused by leakage of the promoter.

We also compared growth with different galactose induction times. As shown in figure 5, samples with equal starting OD600 show faster growth when the galactose induction time is longer. The strain induced for 6 hours grows approximately 2 times faster than the strain induced for 2 hours. To obtain even higher growth rates longer induction can be used, as induction for longer than 6 hours has not been tested.
We attempted growing our strain on cellulose and phosphorylated cellulose, but were unable to detect growth on pure cellulose and phosphorylated cellulose in a plate reader. Absence of growth on pure cellulose is expected as cellulose is notoriously difficult to degrade [1]. However, growth using the cellulosome on phosphorylated cellulose has been shown before [3]. We hypothesized there was no growth due to poor stirring in the plate reader and grew our strain in a flask. Now we were able to grow our strain on phosphorylated cellulose in a flask. A negative control the same strain without carbon source was grown.
Table 1. Growth on phosphorylated cellulose using YPH499 containing PAL2 and the cellulosome (YPH499++). OD600 was measured at t=0, t=12h and t=36h. Initial OD600 value is high due to absorbance from the phosphorylated cellulose.
| OD600 at t=0 | OD600 at t=12h | OD600 at t=36h | |
|---|---|---|---|
| YPHYPH499++ | 0,51 | 0,55 | 1,36 |
| negative control | 0,1 | 0,06 | 0,1 |
Styrene Detection with RP-HPLC UV DAD
After testing PAL2 in E. coli, we tested styrene production in YPH499 with PAL2 using RP-HPLC UV DAD; we based our method on an established protocol [4]. This HPLC separates compounds based on their affinity for the column and measures their absorbance spectrum. A test run gave us the elution conditions and retention time of styrene. Comparing the UV spectrum of the fraction with the known UV spectrum of styrene proved the fraction was indeed styrene.

This resulted in the following HPLC method for styrene detection: using a gradient of methanol and ddH2O with 30% methanol at the start to 100% methanol over 45 minutes. The retention time of styrene under these conditions was determined to be 17,5 min. The strains were grown for two days on 2% glucose and styrene production was boosted with 0,5 g/L phenylalanine (L-phenylalanine). Four different cultures were prepared: a control with phenylalanine (Ctrl), a strain containing PAL2 with phenylalanine (PAL2+), a strain containing PAL2 without phenylalanine (PAL2-) and a strain containing both PAL2 and the cellulosome with phenylalanine (PAL2++). After incubation, the cell culture was centrifuged and the supernatant was loaded onto the column. Samples were also prepared using ethyl acetate extraction, but HPLC runs did not show clear peaks. As such culture supernatant was used for styrene detection.

As can be seen in figure 7 a peak is observed at the retention time of 17,5 min, indicating production of styrene by our cells. Another peak can be observed at 10 minutes retention time. As this peak is not present in the control sample, we hypothesize this is trans-cinnamate. To test this we ran trans-cinnamate on the HPLC system, which indeed showed a peak at the same retention time. The trans-cinnamate peak is considerably larger than the styrene peak. We hypothesize this data indicates that the conversion from trans-cinnamate to styrene by Fdc1 is limiting under these conditions.
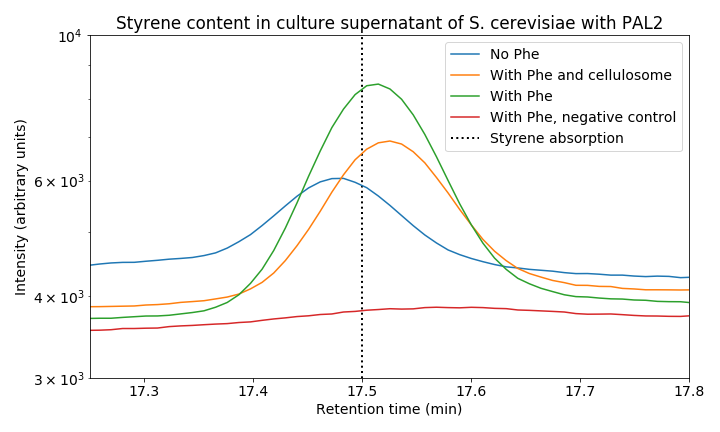
When we zoom in on the styrene peak we can see differences between the tested strains (Figure 8). There is a clear difference between all three PAL2 containing strains compared to the control, indicating successful production of styrene. Both our consolidated bioprocessing strain and our PAL2 containing strain are able to produce styrene. Comparing PAL2 and PAL2+ shows that addition of phenylalanine is not necessary for styrene production; however, it does boosts styrene production indicating that phenylalanine production might be a bottleneck for styrene production. In the table below, an approximation of the produced quantity of styrene can be seen. These results show we have successfully produced styrene in yeast from glucose.
Table 2. Approximation of styrene production per dry weight cells. Method of calculation can be seen here (link to labjournal). Peak area is corrected using a negative control. The OD600 of PAL++ was lower than expected for unknown reasons, which gives an inflated image of the amount of styrene produced by this strain.| OD600 | Peak area (arbitrary units) | styrene/Dryweight cells (ug/mg) | |
|---|---|---|---|
| PAL2 | 4.2 | 17887 | 3.773894298 |
| PAL2+ | 2.41 | 35929 | 13.21081475 |
| PAL2++ | 0.38 | 23768 | 55.4256197 |
Trans-cinnamate and Styrene production from cellobiose
As proof of concept we used our strain containing PAL2 and the cellulosome (YPH499++) to produce styrene. Cells are grown on fructose and induced with galactose for 6 hours, after which the cells are pelleted, washed and transferred to medium containing cellobiose. The same strain grown under the same conditions without cellobiose is taken as a negative control. After growth on cellobiose for 36 hours, styrene production is measured using the RP-HPLC UV DAD as described above. As can be seen from figure 9 small peaks at the retention time of trans-cinnamate and styrene, 10.3 and 17.2 minutes respectively can be observed.

Zooming in on the peak at 10.2 minutes shows more trans-cinnamate is produced when growing on cellobiose in comparison to the negative control (Figure 9). A peak at the retention time of trans-cinnamate was not expected; however, as trans-cinnamate absorption is very strong at 254nm, it is possible contamination of previous samples occurred in the HPLC [6].

When taking a closer look at the peak corresponding to the retention time of styrene, a clear difference can be seen with the control (Figure 11). Although the peak is very small, it is higher than the background of the control. It should be noted that the supernatant of the cell cultured was used for detection of styrene. As styrene has a high affinity for apolar phases, the amount of styrene produced is much larger than can be seen on the HPLC. If time would have permitted it, we would have liked to express the cellulosome constitutively; consistent cellulosome expression should facilitate cellobiose or cellulose degradation and thus increase styrene production.

References
[1] Zhang, Y. P., Cui, J., Lynd, L. R., & Kuang, L. R. (2006). A Transition from Cellulose Swelling to Cellulose Dissolution byo-Phosphoric Acid: Evidence from Enzymatic Hydrolysis and Supramolecular Structure. Biomacromolecules, 7(2), 644-648. doi:10.1021/bm050799c
[2] Ferrari, A. R., Gaber, Y. & Fraaije, M. W. A fast, sensitive and easy colorimetric assay for chitinase and cellulase activity detection. Biotechnol. Biofuels 7, 37 (2014).
[3] Wen, F., Sun, J., & Zhao, H. (2010). Yeast surface display of trifunctional minicellulosomes for simultaneous saccharification and fermentation of cellulose to ethanol. Applied and Environmental Microbiology, 76(4), 1251–60. http://doi.org/10.1128/AEM.01687-09
[4] Mckenna, R., Thompson, B., Pugh, S., & Nielsen, D. R. (2014). Rational and combinatorial approaches to engineering styrene production by Saccharomyces cerevisiae. Microbial Cell Factories, 13(1). doi:10.1186/s12934-014-0123-2
[5] Lei Chen, Zhiwei Xu, Jialu Li, Baoming Zhou, Mingjing Shan, Yinglin Li, Liangsen Liu, Baodong Li and Jiarong Niu: Modifying graphite oxide nanostructures in various media by high-energy irradiation. RSC Adv., 2014, 4, 1025
[6] NIST Chemistry WebBook, SRD 69. (n.d.). Retrieved from https://webbook.nist.gov/cgi/cbook.cgi?ID=C140103&Mask=400#UV-Vis-Spec
