|
|
| Line 7: |
Line 7: |
| | <div class="story-content"> | | <div class="story-content"> |
| | <h2 id="scroll-to">The <span>Results</span></h2> | | <h2 id="scroll-to">The <span>Results</span></h2> |
| − | <p>After we got our purified protein we performed a Western Blot so we could identify it. As we can see in the first photograph our supernatant has our target protein purified because there is a single band. This band only appears when we treat the sample with the Xa protease because if we don’t the protein doesn’t release from the starch granule. In the pellet we have other proteins but we only have our purified protein in the supernatant. When we compared both results with the GFP control we see that the band that appears is at the same level meaning the protein we have purified is also GFP. From one kilogram of fresh weight we extracted 4.8 grams of starch and from them, 120 milligrams of protein.</p> | + | <p><b>1. Plasmid construction using GoldenBraid technology</b></p> |
| − |
| + | <p>In order to build our plasmid, we inserted all the parts that we needed in an empty vector using GoldenBraid technology. The transcriptional unit that we have introduced in our plasmids is formed by the 35S promoter (p35S), the Granule-bound starch synthase (GBSS), a factor Xa cleavage site, our protein of interest in this case the GFP and a terminator (Tnos) (Figure 1).</p> |
| − | <img src="https://static.igem.org/mediawiki/2018/d/de/T--Navarra_BG--results1.jpg" alt="" class="min_height300" style="margin-bottom:30px;"> | + | <div class="text-center mb-20 mb-20"> |
| − |
| + | <img src="https://static.igem.org/mediawiki/2018/8/8f/T--Navarra_BG--results_fig1.jpg" alt="Figure 1. Plasmid including the transcriptional unit GBSS-Xa-GFP." class="max_height350"> |
| − | <img src="https://static.igem.org/mediawiki/2018/7/77/T--Navarra_BG--results2.jpg" alt="" class="min_height300" style="margin-bottom:30px;">
| + | <span class="pie"><strong>Figure 1</strong>. Plasmid including the transcriptional unit GBSS-Xa-GFP.</span> |
| − |
| + | </div> |
| − | <img src="https://static.igem.org/mediawiki/2018/a/ac/T--Navarra_BG--results3.jpg" alt="" class="min_height300" style="margin-bottom:30px;">
| + | |
| | | | |
| − | <p>As it is shown in the graphic, the quantity of starch in plants exposed to volatile compounds is much greater, 10-12 times more. That way instead of getting 120 milligrams of proteins we could get 1200-1440 milligrams of protein.</p> | + | <p><b>2. Transient transformation of <em>Nicotiana benthamiana</em> with the plasmid pGBSS-Xa-GFP.</b></p> |
| | + | <p>After plant transformation we analyzed the present of GFP fluorescent in <em>Nicotina benthamiana</em> leaves using confocal microscopy. As shown in Figure 2 GFP was present in starch granules.</p> |
| | + | <div class="text-center mb-20 mb-20"> |
| | + | <img src="https://static.igem.org/mediawiki/2018/4/43/T--Navarra_BG--results_fig2.jpg" alt="Figure 2. Confocal microscopy of Nicotina benthamiana leaves expressing GBSS-Xa-GFP." class="max_height350"> |
| | + | <span class="pie"><strong>Figure 2</strong>. Confocal microscopy of <em>Nicotina benthamiana</em> leaves expressing GBSS-Xa-GFP.</span> |
| | + | </div> |
| | | | |
| − | <img src="https://static.igem.org/mediawiki/2018/b/ba/T--Navarra_BG--results4.jpg" alt="" class="min_height300"> | + | <p><b>3. Starch purification.</b></p> |
| | + | <p><em>Nicotiana benthamiana</em> transformed leaves were blended, filtrated and centrifuged (Figure 3) to obtain pure starch in a simple, cheap and high efficient way.</p> |
| | + | <div class="text-center mb-20 mb-20"> |
| | + | <img src="https://static.igem.org/mediawiki/2018/5/53/T--Navarra_BG--results_fig3.jpg" alt="Figure 3. Starch purification steps." class="max_height350"> |
| | + | <span class="pie"><strong>Figure 3</strong>. Starch purification steps.</span> |
| | + | </div> |
| | + | |
| | + | <p><b>4. Release of GFP from starch granules.</b></p> |
| | + | <p>Pure starch granules were treated with Xa protease to release free GFP. After proteolytic digestión with Xa protease and a brief centrifugation free GFP was found in the supernatant, while GBSS-GFP remained in pellet fraction as we can see in a western blot using GFP specific antibodies (Figure 4).</p> |
| | + | <div class="text-center mb-20 mb-20"> |
| | + | <img src="https://static.igem.org/mediawiki/2018/8/81/T--Navarra_BG--results_fig4.jpg" alt="Figure 4. Western blot to detect the presence of GFP." class="max_height350"> |
| | + | <span class="pie"><strong>Figure 4</strong>. Western blot to detect the presence of GFP.</span> |
| | + | </div> |
| | + | |
| | + | <p><b>5. Quantification of free GFP.</b></p> |
| | + | <p>To quantify released GFP in our sample we did a western blot using GFP antibodies. We used known amounts of standard GFP and different volumes of our supernatant. To confirm that GFP was only released when Xa protease was present we included a negative control without protease (Figure 5).</p> |
| | + | <div class="text-center mb-20 mb-20"> |
| | + | <img src="https://static.igem.org/mediawiki/2018/d/d8/T--Navarra_BG--results_fig5.jpg" alt="Figure 5. Western blot to quantify GFP." class="max_height350"> |
| | + | <span class="pie"><strong>Figure 5</strong>. Western blot to quantify GFP.</span> |
| | + | </div> |
| | + | <p>We quantified protein bands by densitometry using commercial GFP as control. Our results showed that in this experimental conditions we are able to obtain 0.5 milligrams of pure GFP from 1 kg of leaf fresh weight.</p> |
| | + | |
| | + | |
| | + | <p><b>6. Can we improve GFP yield?</b></p> |
| | + | <p>If we are able to increase the amount of starch in leaves we will increase GFP production. As it is known that volatile compounds emitted by <em>Alternaria alternata</em> increase the starch content in leaves (Sánchez-López et al., 2016), we decided to analyze the increment of starch production in Arabidopsis plants growing in the presence of adjacent <em>Alternaria alternata</em> (Figure 6).</p> |
| | + | <div class="text-center mb-20 mb-20"> |
| | + | <img src="https://static.igem.org/mediawiki/2018/0/0e/T--Navarra_BG--results_fig6.jpg" alt="Figure 6. Volatile Compounds emitted by Alternaria alternata increase the starch content in leaves." class="max_height350"> |
| | + | <span class="pie"><strong>Figure 6</strong>. Volatile Compounds emitted by <em>Alternaria alternata</em> increase the starch content in leaves.</span> |
| | + | </div> |
| | + | <p><em>Alternaria alternata</em> volatiles increased 10 times the leaf starch content, so a production of about 5 milligrams of pure GFP per 1 kg of leaf fresh weight will be expected using this strategy.</p> |
| | </div> | | </div> |
| | </div> | | </div> |
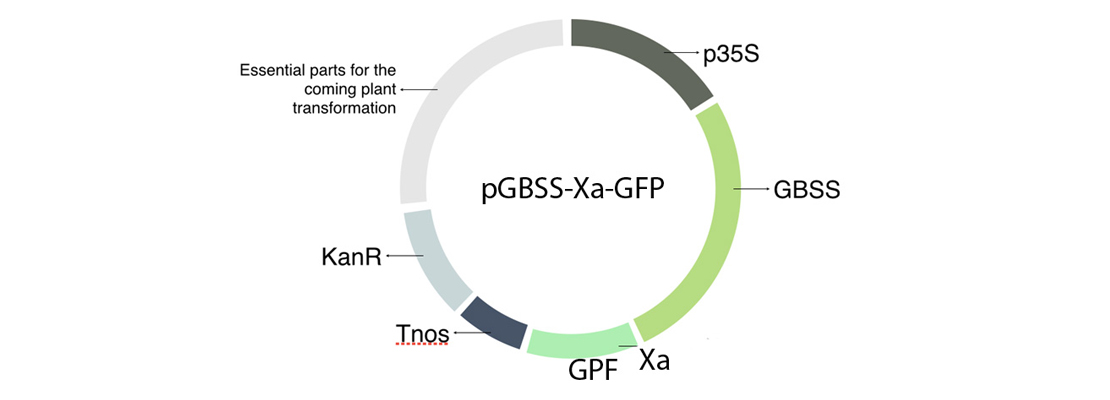 Figure 1. Plasmid including the transcriptional unit GBSS-Xa-GFP.
Figure 1. Plasmid including the transcriptional unit GBSS-Xa-GFP.
 Figure 2. Confocal microscopy of Nicotina benthamiana leaves expressing GBSS-Xa-GFP.
Figure 2. Confocal microscopy of Nicotina benthamiana leaves expressing GBSS-Xa-GFP.
 Figure 3. Starch purification steps.
Figure 3. Starch purification steps.
 Figure 4. Western blot to detect the presence of GFP.
Figure 4. Western blot to detect the presence of GFP.
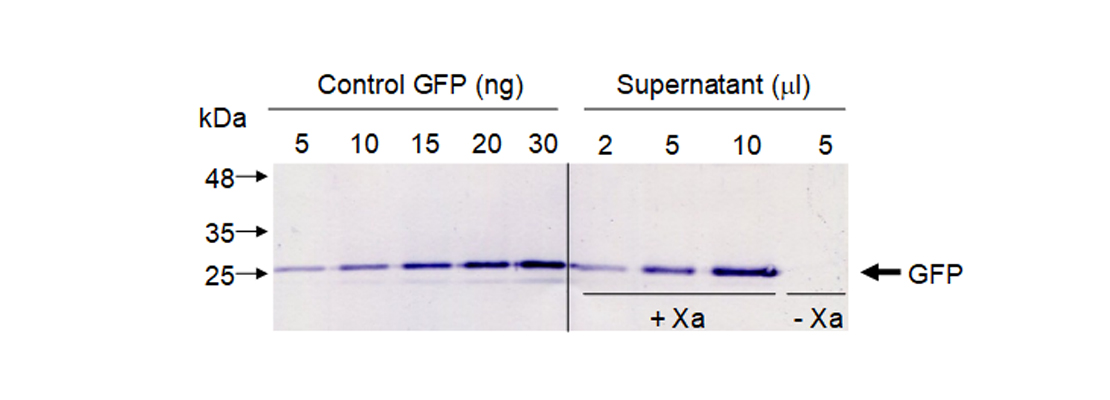 Figure 5. Western blot to quantify GFP.
Figure 5. Western blot to quantify GFP.
 Figure 6. Volatile Compounds emitted by Alternaria alternata increase the starch content in leaves.
Figure 6. Volatile Compounds emitted by Alternaria alternata increase the starch content in leaves.

