

FBA Model for Alkane and MFC
FBA
In the alkane degradation and MFC modules, we adopted Flux Balance Analysis (FBA) model to capture the relationship between different variables and characterize the theoretical maximum values of target outputs. The FBA model is widely used to simulate a genome-wide metabolic network, and the flux distribution between metabolites. With this algorithm, it is possible to maximize an objective function under a set of constraints provided by the user or the genome database, without specific enzyme kinetics inputs. By using FBA, we can analyse the alkane degradation pathway and electron generating reaction while taking into consideration of the complex metabolism network. This algorithm also helped us bypass the enzyme kinetics part, since little has been documented about the ASS enzyme complex in current literature. However, this method was limited to simulate equilibrium state only, and the theoretical limits are highly dependent on the database and constraints provided.[1] In our project, we used iSO783 as Shewanella Oneidensis MR-1 metabolic model, and made adjustment upon it. iSO783 is a widely-used S. Oneidensis MR-1 model containing 774 reactions, and 783 genes. [2]
Hopefully, this mathematical modelling can provide an insight into the interdependence of conditional factors and serve as a guide of our experimental design.
Extracellular Electron Transport
In the MFC, S. oneidensis MR-1 has been reported to transport electrons to electrode in three ways, (1) direct electron transport (DET) mode based on the c-type cytochromes and conductive pili called nanowires, (2) self-secreted flavins to convey electrons and 3) the mediated electron transfer (MET) mode, which relies on exogenous mediators [1]. The DET route, which is a more dominant transport mode without the help of exogenous mediators, is modelled and studied. As reviewed in the MFC part of our wiki, the DET route depends on the c-type cytochrome (MtrC and OmcA) in the cytoplasmic membrane interacting with the electrode. It can also use nanowires to transfer electrons to the electrode that is located distantly from the cells.
In the iSO783, we considered the reaction flux of CYOO2 as our optimization objective as it involved in the reduction of a type of cytochrome c protein, denoted as Cco (SO2361 and SO2362 and SO2363 and SO2364) or Cyco (SO4606 and SO4607 and SO4609) in iSO783). In such case, the reaction flux of CYOO2 is taken to estimate the DET flux.
DET- Lactate
As noticed the ability of S. oneidensis MR-1 utilizing lactate to generate electricity [3], we first examined the FBA model using lactate as the carbon source for electricity generation. We examined the effect of varying lactate uptake flux on two separate objectives, i.e. maximizing biomass growth and maximizing DET flux. We assume 5% of maximum growth as the boundary of biomass growth flux, which would be the minimum viable growth rates in practice as described by Mao [4].
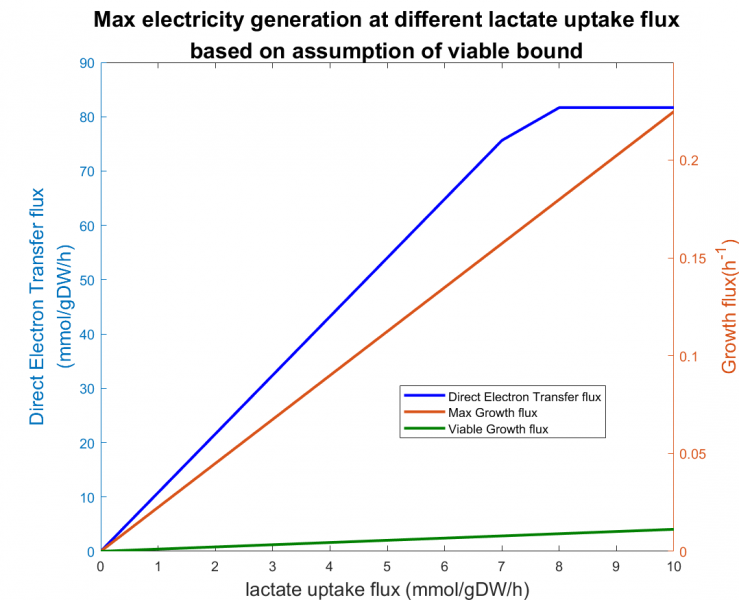
Figure 4. The effect of lactate on maximum direct electron transfer(DET) flux of Shewanella utilizing lactate at viable growth flux boundary
Figure 4 shows that the DET flux reached a plateau at 81.68 mmol/gDW/h as lactate uptake flux increased. An upper bound existed such that increasing lactate uptake would no longer increase the electricity generation.

Figure 5. The trade off relationship between direct electron transfer(DET) flux and biomass growth flux when lactate uptake is set as constant
Noted that the biomass growth is driven to its lower bound when the optimization objective is set as DET flux, we studied the relationship between DET flux with biomass flux at constant lactate input. In figure 5, the DET flux first sustained at 81.68 mmol/gDW/h but then decreased when biomass flux increased beyond 0.164h-1. This implies the electricity generation could possibly be maximized when below a certain growth rate, however, higher growth rate could possibly decrease the electricity generation.
Shewanella Growth in Hexane

Figure 6. Maximum growth flux of Shewanella uptaking hexane under aerobic and anaerobic condition
Figure 6 shows the maximized growth flux generated by FBA model for aerobic and anaerobic conditions. Aerobic condition is set as oxygen flux=0, fumarate flux lower bound=-20.42mmol/gDW/h,acetate flux upper bound=3.134mmol/gDW/h, pyruvate flux upper bound=0.872mmol/gDW/h, and hexane flux ranges from -5mmol/gDW/h to zero. Aerobic condition is set as oxygen flux lower bound=20.42mmol/gDW/h
In this part, we used FBA to generate the maximized growth flux of Shewanella oneidensis MR-1 when providing hexane as energy source. Reaction constraints were set based on literature [5] values and genome database was adjusted to suit our engineered construct by adding the alkane degradation reactions. Results show that the maximized growth under aerobic condition is around 0.513 h-1 , while the anaerobic growth flux is around 0.298h-1 , both enough to sustain growth. This means theoretically, Shewanella oneidensis MR-1 is able to grow in a hexane-containing media without provision of lactate or glucose under both aerobic and anaerobic conditions.
Maximum current generation and power output
Current is given by the derivative of the electric charge over time. The electron flux can be converted to current using Faraday’s constant 96485 C/mol

Given the maximum DET flux is 81.68mmol/gDW/h, therefore, the Shewanella oneidensis MR-1 is possible to generate current up to 2.189A/gDW.
The upper limit of MFC cell voltage is calculated based on the difference of standard cell potential in the anode and cathode. The standard potential are summarized in the table.
|
|
Redox couple |
Eo (V) |
|
Anode |
Cytochrome c (Fe3+) + e− → Cytochrome c (Fe2+) |
+0.254[6] |
|
Cathode |
Ferricyanide [Fe(CN)6]3− + e− → Ferrocyanide [Fe(CN)6]4− |
+0.436[7] |
|
|
O2 + 4H++4e− → 2H2O |
+0.51 [8] |
In the MFC design that uses ferricyanide as cathode, the maximum cell voltage would be 0.182V. If oxygen is used as cathode, the maximum cell voltage would be 0.256V. The maximum power output could be 0.398W/gDW in ferricyanide while 0.56W/gDW could be reached using oxygen as cathode.
Effect of fumarate

Figure 7. The effect of fumarate on maximum direct electron transfer(DET) flux of Shewanella utilizing hexane at viable growth flux boundary
In pursuit of an overall higher electron output, we tried to identify how the nutritional factors in culturing media affect DET flux and growth flux. Hexane concentration was found to play a minor role since the actual uptake flux was restricted to be low(around -6.04*10-7 mmol/gDW/h) by internal factors of the bacteria.
However, fumarate was found to be an essential factor since it affects for growth, DET, and hexane uptake. In figure 7, the blue curve shows a positive correlation between fumarate uptake and DET flux (with growth flux set to 5%), and DET achieves its maximum value 81.68mmol/gDW/h at a fumarate uptake flux of 8 mmol/gDW/h. The green curve is the corresponding growth flux when DET was optimized. Since DET and growth cannot be optimized simultaneously, we separately constructed the red curve to describe the relationship between growth flux and fumarate uptake when optimizing growth. The curve shows a positive correlation and growth flux achieves its maximum value 0.9408h-1 when fumarate uptake flux reaches 94 mmol/gDW/h.
Aeration effect on DET flux
We also explored the difference of DET flux under aerobic and anaerobic condition. In anaerobic condition, we assumed fumarate as the electron acceptor. It is surprising that the DET flux is driven to extremely small negative value under anaerobic condition, while DET flux can reach 81.68mmol/gDW/h when oxygen flux is set as 20.42mmol/gDW/h according to literature[4].
REFERENCES
- Orth, J.D., Thiele, I. and Palsson, B.(2010). What is Flux Balance Analysis? Nature Biotechnology, [1112-9778], Volume:28, Issue:3, Page:245, doi:10.1038/nbt.1614 Available at https://www.nature.com/articles/nbt.1614
- 2012.igem.org. (2015). Team: NTU-Singapore/Modeling. [online] Available at: https://2015.igem.org/Team:NTU-Singapore/Modeling
- L. Mao and W. Verwoerd, "Theoretical exploration of optimal metabolic flux distributions for extracellular electron transfer by Shewanella oneidensis MR-1", Biotechnology for Biofuels, vol. 7, no. 1, 2014.
- G. Newton, S. Mori, R. Nakamura, K. Hashimoto and K. Watanabe, "Analyses of Current-Generating Mechanisms of Shewanella loihica PV-4 and Shewanella oneidensis MR-1 in Microbial Fuel Cells", Applied and Environmental Microbiology, vol. 75, no. 24, pp. 7674-7681, 2009.
- L. Berthe-Corti and W. Ebenhöh, "A mathematical model of cell growth and alkane degradation in Wadden Sea sediment suspensions", Biosystems, vol. 49, no. 3, pp. 161-189, 1999.
- B. Virdis, S. Freguia, R. Rozendal, K. Rabaey, Z. Yuan and J. Keller, "Microbial Fuel Cells", Treatise on Water Science, pp. 641-665, 2011.
- M. Pandurangachar, B. Kumara Swamy, B. Chandrashekar, Ongera Gilbert, Sathish Reddy and B. Sherigara, "Electrochemical Investigations of Potassium Ferricyanide and Dopamine by 1-butyl-4-methylpyridinium tetrafluoro borate Modified Carbon Paste Electrode: A Cyclic Voltammetric Study",International Journal of Electrochemical Science, vol. 5, no. 8, pp. 1187-1202, 2010.
- U. Schröder, "Anodic electron transfer mechanisms in microbial fuel cells and their energy efficiency", Phys. Chem. Chem. Phys., vol. 9, no. 21, pp. 2619-2629, 2007.
1

