| Line 403: | Line 403: | ||
<i class="icon-paint-brush"></i> | <i class="icon-paint-brush"></i> | ||
<img src="https://static.igem.org/mediawiki/2018/d/dd/T--Newcastle--backboneamplification.jpeg"> | <img src="https://static.igem.org/mediawiki/2018/d/dd/T--Newcastle--backboneamplification.jpeg"> | ||
| − | <p | + | <p class="text-align:center"><br> Figure 2: Agarose gel showing the 2kb band representing the amplified plasmid backbone, a ladder for 1kb hyperladder from Bioline is shown that was used throughout.</p> |
</div> | </div> | ||
</div> | </div> | ||
Revision as of 11:02, 15 October 2018
Alternative Roots
Naringenin Operon Assembly Results
Results
Introduction
An operon containing four genes encoding the naringenin biosynthetic pathway (Figure 1) was previously assembled by TU Darmstadt 2014 iGEM team (BBa_K1497016). We needed to assemble a naringenin operon to allow the genes that encode for the enzymes that produce naringenin to be implemented into DH5 alpha cells. Modifications were made to TU Darmstadt’s operon, as the second gene in the operon (TAL) needed to be manually codon optimised to be synthesised by IDT. Further the last two genes CHS and CHI in the operon were switched in the Benchling design for our operon. These gblocks were ordered from IDT with the relevant primers for amplification and detection of the gblocks. We decided to use Gibson Assembly, because it was how TU Darmstadt assembled their operon successfully and the assembly is a scarless method based on the overlaps of fragments.
The first part of this to accomplish was amplifying the pSB1C3 backbone that would take up the naringenin operon. Competent cells were then to be produced and tested using a positive control for Gibson Assembly. The gblocks synthesised by IDT were utilised in Gibson Assemblies and subsequent transformation of the naringenin operon into DH5 alpha competent cells was to be attempted. This would be tested through a series of minipreps and sequencing of transformed colonies. Alongside this a new design of the naringenin operon based on the pathway modelling, was constructed to show how different promoters with varying strengths could increase naringenin production.

Figure 1: The naringenin synthesis pathway from L-tyrosine.
Results
Experimental Work
Plasmid Design
The naringenin operon contains the genes for four enzymes: 4 – Coumaryl ligase, Tyrosine ammonia lyase, Chalcone isomerase and Chalcone synthase. Each of these genes will contain a strong ribosome binding site and has an Escherichia coli his operon terminator. This construct will be under the control of a J23100 constitutive promoter to observe its expression in E.coli as a proof of concept. Once biosynthesis under the control of J23100 is achieved, the construct future experiments would test this under a T7 promoter in E. coli. Parts for biosynthesis in the final chassis organism, root-colonising Pseudomonas sp., will be constructed in the plasmid backbone pSB1C3.
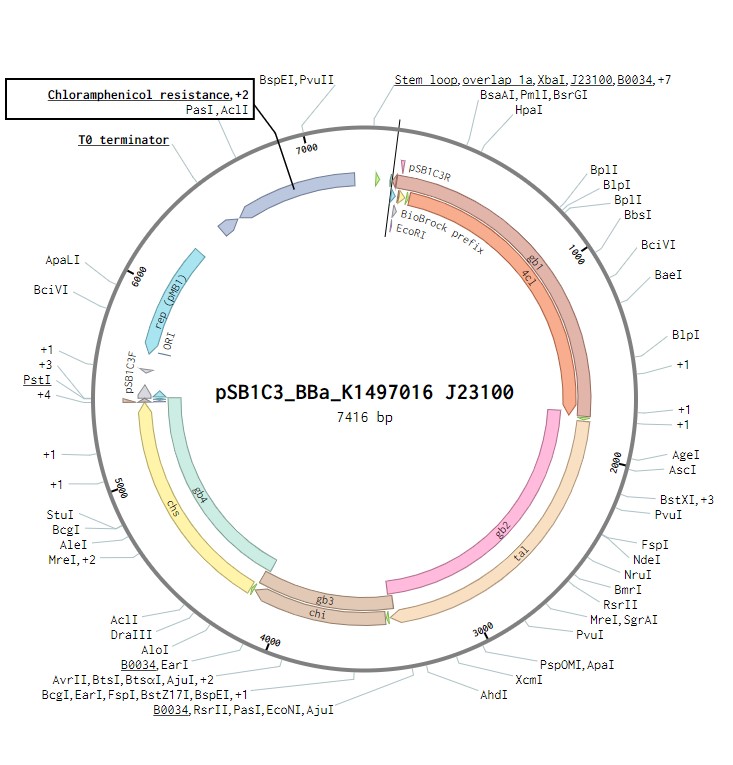
The naringenin biosynthetic operon under control of a J23100 promoter created in Benchling.

The naringenin biosynthetic operon under control of a T7 promoter created in Benchling.

The naringenin biosynthetic operon construct under control of a J23100 promoter created in SBOL.

The naringenin biosynthetic operon contruct under control of a T7 promoter created in SBOL.
Results of the naringenin pathway modelling showed that weaker expression of the first two genes and 10 fold stronger expression of the last two genes would reduce the build-up of malonyl CoA and optimise naringenin synthesis. Therefore we found two synthetic promoters: BG28 and BG51 and an additional E. coli his operon terminator to be placed after the first two genes. These will allow enhanced naringenin production in future experiments, as BG28 is a weak promoter for the first two genes and BG51 is a strong promoter for the last two.
The naringenin biosynthetic operon under control of synthetic promoters BG28 and BG51 created in Benchling.

A close up of the synthetic promoters BG28 and BG51 placement in the operon, created in Benchling.

The naringenin biosynthetic operon contruct under control of a BG28 and BG51 promoters and an additional his operon terminator created in SBOL.
Table 1: Primers designed in Benchling for amplification and detection of the 4 gblocks and the pSB1C3 backbone.
| Primer name | Sequence | Tm | Ta Q5 | Amplified product(bp) | Shown to work | Description |
|---|---|---|---|---|---|---|
| pSB1C3F | tactagtagcggccgctgc | 70 | 71 | 2070 | 6/8/18 | To amplify pSB1C3 backbone |
| pSB1C3R | ctctagaagcggccgcga | 70 | 71 | 2070 | 6/8/18 | To amplify pSB1C3 backbone |
| 4CLF | ccaaatcgccgccaattttc | 59 | 56 | 1686 | 6/9/18 | To detect 4CL part |
| 4CLR | cgtcgtcgttttgaagtggt | 59.07 | 56 | 1686 | 28/9/18 | To detect 4CL part |
| TALF | gaatgtccgaacgctacagg | 58.72 | 55 | 1649 | 28/9/18 | To detect TAL part |
| TALR | tcggaattgagcaggtcgat | 59.18 | 56 | 1649 | 28/9/18 | To detect TAL part |
| CHIF | ctgggcatagaggtctggag | 58.95 | 56 | 726 | 28/9/18 | To detect CHI part |
| CHIR | caccttctccgagtactgct | 58.82 | 56 | 726 | 28/9/18 | To detect CHI part |
| CHSF | aagacgtgcctgggttgata | 59.02 | 56 | 1197 | 28/9/18 | To detect CHS part |
| CHSR | gcttctcctccttcaaccct | 59.01 | 56 | 1197 | 6/9/18 | To detect CHS part |
| gb1F | ctggaattcgcggccgct | 72 | 54 | 1686 | 20/9/18 | To amplify 4CL part |
| gb1R | ttacaatccatttgctag | 53 | 54 | 1686 | 20/9/18 | To amplify 4CL part |
| gb2F | ggcaaaactagcaaatgg | 59 | 59 | 1649 | 20/9/18 | To amplify TAL part |
| gb2R | ttatcagacgggagattg | 58 | 59 | 1649 | 20/9/18 | To amplify TAL part |
| gb3F | cttgcagcaatctcccgt | 65 | 59 | 726 | 20/9/18 | To amplify CHI part |
| gb3R | ctagactccaatcactgg | 58 | 59 | 726 | 20/9/18 | To amplify CHI part |
| gb4F | tactattccagtgattgg | 54 | 55 | 1197 | 20/9/18 | To amplify CHS part |
| gb4R | cggactgcagcggccgct | 78 | 55 | 1197 | 20/9/18 | To amplify CHS part |
The backbone was amplified, purified and quantified to prepare for Gibson assembly, its stock concentration was found to be 26.2 µg/ ml. This would allow the overlapping ends of the gblocks to ligate to the plasmid backbone. The amplification was done by PCR, purified using QIAquick PCR Purification Kit (250) and quantified by use of a Qubit fluorometer. Details of these protocols can be found here…..
Results
Conclusions
Two colonies from Gibson transformations resulted in a 7kb band, corresponding to the size of the operon and plasmid backbone. However sequencing was inconclusive therefore this operon could not be classified as a working part. Future experimentation should attempt to assemble the gblocks one by one into the plasmid backbone and gain sequencing results that show full alignment. Following this the 7kb plasmid should be transformed into BL21 expression cells to produce naringenin, to be extracted using ethyl acetate and measured through HPLC. HPLC of stock naringenin should be conducted for comparison. Observing the expression of the operon using T7 promoters instead of the J23100 constitutive promoter, to see if naringenin production is enhanced. This could be implemented using T7 primers and Q5 site directed mutagenesis in E. coli.



