Tugbainanc (Talk | contribs) |
|||
| (5 intermediate revisions by 2 users not shown) | |||
| Line 23: | Line 23: | ||
<h4>What are furfural and 5-hydroxymethylfurfural?</h4> | <h4>What are furfural and 5-hydroxymethylfurfural?</h4> | ||
| − | <div class="col-md-6"> | + | <div class="col-md-6" style="text-align: center"> |
<img src="https://static.igem.org/mediawiki/2018/b/bd/T--METU_HS_Ankara--de02.jpg" /> | <img src="https://static.igem.org/mediawiki/2018/b/bd/T--METU_HS_Ankara--de02.jpg" /> | ||
<i class="parts-info"> | <i class="parts-info"> | ||
| Line 30: | Line 30: | ||
</div> | </div> | ||
| − | <div class="col-md-6"> | + | <div class="col-md-6" style="text-align: center"> |
<img src="https://static.igem.org/mediawiki/2018/e/e9/T--METU_HS_Ankara--de03.jpg" /> | <img src="https://static.igem.org/mediawiki/2018/e/e9/T--METU_HS_Ankara--de03.jpg" /> | ||
<i class="parts-info"> | <i class="parts-info"> | ||
| Line 42: | Line 42: | ||
<p> | <p> | ||
| − | A corresponding compound, 5-hydroxymethylfurfural (5-HMF), is formed of hexose sugars such as glucose and fructose (Kumar & Sharma, 2017; Wang et al., 2012). | + | A corresponding compound, 5-hydroxymethylfurfural (5-HMF), is formed of hexose sugars such as glucose and fructose (Kumar & Sharma, 2017; Wang <i>et al.</i>, 2012). |
</p> | </p> | ||
| Line 48: | Line 48: | ||
These toxic furans are known to suppress cell growth, decrease ethanol yield, cause damages in the cell DNA and | These toxic furans are known to suppress cell growth, decrease ethanol yield, cause damages in the cell DNA and | ||
harm many enzymes during glycolysis, therefore, pose a great danger to the convenience of lignocellulosic ethanol | harm many enzymes during glycolysis, therefore, pose a great danger to the convenience of lignocellulosic ethanol | ||
| − | production (Ask et al., 2013). These toxins are proven to affect not only E.coli KO11 that was used in our project | + | production (Ask <i>et al.</i>, 2013). These toxins are proven to affect not only E.coli KO11 that was used in our project |
but also other ethanologenic organisms such as <i>Saccharomyces cerevisiae</i> a strain of yeast, which is widely used in | but also other ethanologenic organisms such as <i>Saccharomyces cerevisiae</i> a strain of yeast, which is widely used in | ||
| − | fermentation. (Wang et al., 2012; Almeida et al., 2007; Ask et al., 2013). | + | fermentation. (Wang <i>et al.</i>, 2012; Almeida <i>et al.</i>, 2007; Ask <i>et al.</i>, 2013). |
</p> | </p> | ||
<p> | <p> | ||
Moreover, the addition of furfural to hemicellulose hydrolysates has been shown to restore toxicity | Moreover, the addition of furfural to hemicellulose hydrolysates has been shown to restore toxicity | ||
| − | (Wang et al., 2011). Furfural is, hence, unique in potentiating the toxicity of other compounds unlike | + | (Wang <i>et al.</i>, 2011). Furfural is, hence, unique in potentiating the toxicity of other compounds unlike |
| − | various hydrolysate inhibitors, hindering the bioethanol production drastically (Wang et al., 2012). | + | various hydrolysate inhibitors, hindering the bioethanol production drastically (Wang <i>et al.</i>, 2012). |
</p> | </p> | ||
<h3>Our solution: Simultaneous expression of FucO and GSH</h3> | <h3>Our solution: Simultaneous expression of FucO and GSH</h3> | ||
| − | <h4>FucO :L-1,2-Propanediol Oxidoreductase</h4> | + | <h4>FucO: L-1,2-Propanediol Oxidoreductase</h4> |
<p> | <p> | ||
| Line 72: | Line 72: | ||
<p> | <p> | ||
| − | This suggest that YqhD will also have detrimental effects on the growth and living of the bacteria as it will compete with the biosynthesis of NADPH during fermentation in E.coli (Zheng, 2013). The native conversion of NADH to NADPH in E. coli is already insufficient to revitalize the NADPH pool during fermentation, so the actions shouldn’t be interfering with NADPH metabolism (Wang et al., 2011; 2012). | + | This suggest that YqhD will also have detrimental effects on the growth and living of the bacteria as it will compete with the biosynthesis of NADPH during fermentation in E.coli (Zheng, 2013). The native conversion of NADH to NADPH in <i>E. coli</i> is already insufficient to revitalize the NADPH pool during fermentation, so the actions shouldn’t be interfering with NADPH metabolism (Wang <i>et al.</i>, 2011; 2012). |
</p> | </p> | ||
| Line 79: | Line 79: | ||
</p> | </p> | ||
| − | <img src="https://static.igem.org/mediawiki/2018/1/1a/T--METU_HS_Ankara--de04.jpg" /> | + | <div class="col-md-12" style="text-align: center"> |
| − | + | <img width="700" src="https://static.igem.org/mediawiki/2018/1/1a/T--METU_HS_Ankara--de04.jpg" /> | |
| − | + | <div class="clear: both"></div> | |
| − | + | <i class="parts-info"> | |
| + | Figure 3: Possible reaction pathways for furfural hydrogenation | ||
| + | </i> | ||
| + | </div> | ||
<h4>GSH: Bifunctional gamma-glutamate-cysteine ligase/Glutathione synthetase</h4> | <h4>GSH: Bifunctional gamma-glutamate-cysteine ligase/Glutathione synthetase</h4> | ||
<p> | <p> | ||
| − | In normal circumstances, simultaneous detoxification of ROS (Reactive Oxygen Species), the conversion of HMF and furfural drag the cell into a more oxidized intracellular environment and deteriorate the antioxidant defense system of the cell (Ask et al., 2013). Here, increasing the cellular levels of antioxidants are essential. | + | In normal circumstances, simultaneous detoxification of ROS (Reactive Oxygen Species), the conversion of HMF and furfural drag the cell into a more oxidized intracellular environment and deteriorate the antioxidant defense system of the cell (Ask <i>et al.</i>, 2013). Here, increasing the cellular levels of antioxidants are essential. |
</p> | </p> | ||
<p> | <p> | ||
| − | In our case the expression of glutathione (GSH) stands the best chance, as it is the master antioxidant in living organisms coping with ROS, oxidative stress and increasing furan tolerance (Kim & Hahn, 2013; Höck et al., 2013). | + | In our case the expression of glutathione (GSH) stands the best chance, as it is the master antioxidant in living organisms coping with ROS, oxidative stress and increasing furan tolerance (Kim & Hahn, 2013; Höck <i>et al.</i>, 2013). |
</p> | </p> | ||
<p> | <p> | ||
| − | Additionally, a GSH derivative, glutathione S-transferase, reacts with a number of harmful chemical species; such as ROS, reactive nitrogen species (RNS) and xenobiotics including environmental carcinogens (Dasari et al., 2017). | + | Additionally, a GSH derivative, glutathione S-transferase, reacts with a number of harmful chemical species; such as ROS, reactive nitrogen species (RNS) and xenobiotics including environmental carcinogens (Dasari <i>et al.</i>, 2017). |
</p> | </p> | ||
<p> | <p> | ||
| − | In consideration of these studies, we have come to the conclusion that, the concurrent overexpression of GSH and FucO would increase cellular growth rates, lifespan and ethanol production yield (Ask et al., 2013). | + | In consideration of these studies, we have come to the conclusion that, the concurrent overexpression of GSH and FucO would increase cellular growth rates, lifespan and ethanol production yield (Ask <i>et al.</i>, 2013). |
</p> | </p> | ||
| Line 108: | Line 111: | ||
</p> | </p> | ||
| − | <img src="https://static.igem.org/mediawiki/2018/d/dc/T--METU_HS_Ankara--cparts01256eie6.jpg" /> | + | <div class="col-md-12" style="text-align: center"> |
| − | + | <img src="https://static.igem.org/mediawiki/2018/d/dc/T--METU_HS_Ankara--cparts01256eie6.jpg" /> | |
| − | + | <div class="clear: both"></div> | |
| − | + | <i class="parts-info"> | |
| + | Figure 4: Representation of our gene circuit. | ||
| + | </i> | ||
| + | </div> | ||
<p> | <p> | ||
| − | The first protein coding region we have, placed after the RBS, FucO, codes for L-1,2-propanediol oxidoreductase (a homodimer enzyme) in order to act upon furfural presence in the field (Zheng, 2013). The metabolism of furfural by NADH-dependent oxidoreductases reduces the toxicity of the chemical by turning it into furfuryl alcohol, a derivative (Zheng, 2013; Wang et al., 2013; Allen et al., 2010). | + | The first protein coding region we have, placed after the RBS, FucO, codes for L-1,2-propanediol oxidoreductase (a homodimer enzyme) in order to act upon furfural presence in the field (Zheng, 2013). The metabolism of furfural by NADH-dependent oxidoreductases reduces the toxicity of the chemical by turning it into furfuryl alcohol, a derivative (Zheng, 2013; Wang <i>et al.</i>, 2013; Allen <i>et al.</i>, 2010). |
</p> | </p> | ||
| Line 122: | Line 128: | ||
<p> | <p> | ||
| − | GSH is usually found in the thiol-reduced form in living organisms which is crucial for the detoxification of reactive oxygen species and the other free radicals (Ask et al., 2013). | + | GSH is usually found in the thiol-reduced form in living organisms which is crucial for the detoxification of reactive oxygen species and the other free radicals (Ask <i>et al.</i>, 2013). |
</p> | </p> | ||
<p> | <p> | ||
| − | Reactive oxygen species (ROS) are harmful substances that alter protein based matters by interacting with their electrons as themselves have an unbalanced electron state (Lu, 2013; Burton & Jauniaux, 2011). Because many benefits of GSH include scavenging of ROS, protection against endogenous toxic metabolites and detoxification of xenobiotics, we chose this gene to include in our bacteria (Höck et al., 2013). | + | Reactive oxygen species (ROS) are harmful substances that alter protein based matters by interacting with their electrons as themselves have an unbalanced electron state (Lu, 2013; Burton & Jauniaux, 2011). Because many benefits of GSH include scavenging of ROS, protection against endogenous toxic metabolites and detoxification of xenobiotics, we chose this gene to include in our bacteria (Höck <i>et al.</i>, 2013). |
</p> | </p> | ||
| Line 197: | Line 203: | ||
<p> | <p> | ||
| − | For the fermentation processes, pressed and dried quince pomace that is composed of 28.8% glucose, 55.7% fructose and 10.1% sucrose based on dry mass was used instead of glucose (Deniz et al | + | For the fermentation processes, pressed and dried quince pomace that is composed of 28.8% glucose, 55.7% fructose and 10.1% sucrose based on dry mass was used instead of glucose (Deniz <i>et al.</i> 2015). After quince pomace and LB were mixed aseptically, fermentation was carried out at pH 6 with automatic KOH additions in the field to avoid pH levels lower than 6. Solids were separated via centrifugation at 5000 g for 5 min at 5°C and supernatant was used to calculate ethanol and reduced sugar. (Deniz <i>et al.</i> 2015) |
</p> | </p> | ||
<p> | <p> | ||
| − | Total soluble reducing sugar content of quince pomace was obtained by dinitrosalicylic acid (DNS) method in which absorbance was measured at 540 nm and ethanol concentrations of test groups were determined by using a Gas Chromatograph. (Deniz et al | + | Total soluble reducing sugar content of quince pomace was obtained by dinitrosalicylic acid (DNS) method in which absorbance was measured at 540 nm and ethanol concentrations of test groups were determined by using a Gas Chromatograph. (Deniz <i>et al.</i> 2015) |
</p> | </p> | ||
| Line 207: | Line 213: | ||
<p> | <p> | ||
| − | By modifying E. coli strain KO11, we have actually designed bacteria that can cope with reactive oxygen species, free radicals and furan byproducts without interfering with the NADPH metabolism (Höck et al., 2013; Wang et al., 2011). | + | By modifying <i>E. coli</i> strain KO11, we have actually designed bacteria that can cope with reactive oxygen species, free radicals and furan byproducts without interfering with the NADPH metabolism (Höck <i>et al.</i>, 2013; Wang <i>et al.</i>, 2011). |
</p> | </p> | ||
<p> | <p> | ||
| − | Thus, our parts would be also feasible in other conditions in which toxins such as ROS or inhibitors such as heavy metals exist (Ask et al., 2013; Wang et al., 2011). This genetical improvement is due to GSH, which is an antioxidant that detoxifies free radicals and such types; and FucO which is an oxidoreductase that transforms HMF and furfural to non-toxic alcohols without getting in the way of the bacteria’s own metabolism (Ask et al., 2013; Wang et al., 2011). | + | Thus, our parts would be also feasible in other conditions in which toxins such as ROS or inhibitors such as heavy metals exist (Ask <i>et al.</i>, 2013; Wang <i>et al.</i>, 2011). This genetical improvement is due to GSH, which is an antioxidant that detoxifies free radicals and such types; and FucO which is an oxidoreductase that transforms HMF and furfural to non-toxic alcohols without getting in the way of the bacteria’s own metabolism (Ask <i>et al.</i>, 2013; Wang <i>et al.</i>, 2011). |
</p> | </p> | ||
| Line 220: | Line 226: | ||
| − | |||
<div class="container"> | <div class="container"> | ||
<div class="row"> | <div class="row"> | ||
| Line 358: | Line 363: | ||
</div> | </div> | ||
</div> | </div> | ||
| − | |||
</div> | </div> | ||
Latest revision as of 14:33, 17 October 2018



 Figure 1: Chemical structure of furfural
Figure 1: Chemical structure of furfural
 Figure 2: Chemical structure of 5-HMF
Figure 2: Chemical structure of 5-HMF
 Figure 3: Possible reaction pathways for furfural hydrogenation
Figure 3: Possible reaction pathways for furfural hydrogenation
 Figure 4: Representation of our gene circuit.
Figure 4: Representation of our gene circuit.
 Figure 5: Representation of our experimental plan/ assay no. 1.
Figure 5: Representation of our experimental plan/ assay no. 1.
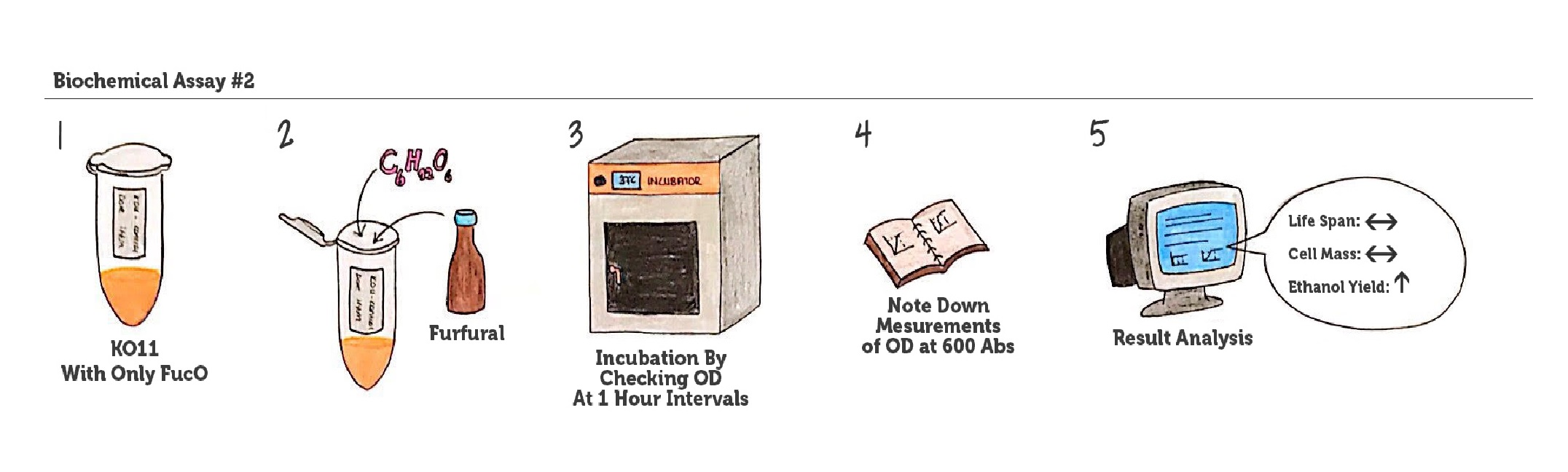 Figure 6: Representation of our experimental plan/ assay no. 2.
Figure 6: Representation of our experimental plan/ assay no. 2.
 Figure 7: Representation of our experimental plan/ assay no. 3.
Figure 7: Representation of our experimental plan/ assay no. 3.
 Figure 8: Representation of our experimental plan/ assay no. 4.
Figure 8: Representation of our experimental plan/ assay no. 4.