Huiyee11186 (Talk | contribs) |
|||
| (One intermediate revision by one other user not shown) | |||
| Line 100: | Line 100: | ||
<p class="pcontent">Ribulose-1,5-biphosphate carboxylase/oxygenase is one of the world’s most abundant enzyme. | <p class="pcontent">Ribulose-1,5-biphosphate carboxylase/oxygenase is one of the world’s most abundant enzyme. | ||
It catalyzes the conversion of inorganic carbon into organic carbon. | It catalyzes the conversion of inorganic carbon into organic carbon. | ||
| − | In our designed pathway, the function of the Rubisco is to convert | + | In our designed pathway, the function of the Rubisco is to convert ribulose-1,5-biphosphate (RuBP) from the upper pathway and carbon dioxide into 3-phosphoglycerate (3PGA). |
3PGA will then be converted to pyruvate by the native metabolic system of <i>E. coli</i>. | 3PGA will then be converted to pyruvate by the native metabolic system of <i>E. coli</i>. | ||
After mining information from various publications, | After mining information from various publications, | ||
| − | we selected Rubisco from <i>Synechococcus elongatus</i> | + | we selected Rubisco from <i>Synechococcus elongatus</i> PCC 7002, which is a well-studied cyanobacteria. |
Its genome is completely sequenced and it is often used as a model organism for gene manipulation. | Its genome is completely sequenced and it is often used as a model organism for gene manipulation. | ||
Previous research has utilized <i>E. coli</i> as a host of random mutagenesis to enhance the activity of <i>Synechococcus</i> Rubisco. | Previous research has utilized <i>E. coli</i> as a host of random mutagenesis to enhance the activity of <i>Synechococcus</i> Rubisco. | ||
| Line 131: | Line 131: | ||
<p class="pcontent">Measurement of 3PGA or pyruvate concentration could not directly reflect the activity of Rubisco | <p class="pcontent">Measurement of 3PGA or pyruvate concentration could not directly reflect the activity of Rubisco | ||
since both of them are important metabolites that will flow to downstream metabolic pathway. | since both of them are important metabolites that will flow to downstream metabolic pathway. | ||
| − | We then decided to determine its function by total solution test which we will mention below. | + | We then decided to determine its function by a total solution test which we will mention below. |
</p> | </p> | ||
</div> | </div> | ||
| Line 140: | Line 140: | ||
Oxygen competes with CO<sub>2</sub> as a substrate for Rubisco, giving rise to photorespiration. | Oxygen competes with CO<sub>2</sub> as a substrate for Rubisco, giving rise to photorespiration. | ||
To overcome this problem, some photosynthetic organisms have evolved their own carbon concentrating | To overcome this problem, some photosynthetic organisms have evolved their own carbon concentrating | ||
| − | + | mechanism (CCM), which helps to maintain a sufficient amount of CO<sub>2</sub> around Rubisco. | |
</p> | </p> | ||
<p class="pcontent">We are inspired by the carbon concentrating mechanisms (CCM) of cyanobacteria. | <p class="pcontent">We are inspired by the carbon concentrating mechanisms (CCM) of cyanobacteria. | ||
In cyanobacteria, Rubisco and carbonic anhydrase (CA) is encapsulated in a microcompartment, the carboxysome. | In cyanobacteria, Rubisco and carbonic anhydrase (CA) is encapsulated in a microcompartment, the carboxysome. | ||
Carbonic anhydrase, also known as carbonate dehydratase, is involved in the interconversion between CO<sub>2</sub> and HCO<sub>3</sub><sup>-</sup>. This enzyme can be found in most organisms, including <i>E. coli</i> but the difference is its catalyzing rate in hydration and dehydration of CO2. Therefore, | Carbonic anhydrase, also known as carbonate dehydratase, is involved in the interconversion between CO<sub>2</sub> and HCO<sub>3</sub><sup>-</sup>. This enzyme can be found in most organisms, including <i>E. coli</i> but the difference is its catalyzing rate in hydration and dehydration of CO2. Therefore, | ||
| − | we will incorporate into our system the carbonic anhydrase gene from <i>Synechococcus elongatus</i> | + | we will incorporate into our system the carbonic anhydrase gene from <i>Synechococcus elongatus</i> PCC 7002. |
</p> | </p> | ||
<img class="gif" src="https://static.igem.org/mediawiki/2018/3/34/T--NCKU_Tainan--design_CA.gif" alt="Rubisco"> | <img class="gif" src="https://static.igem.org/mediawiki/2018/3/34/T--NCKU_Tainan--design_CA.gif" alt="Rubisco"> | ||
| Line 167: | Line 167: | ||
<p class="pcontent">To measure the enzyme activity of CA, we compare the conversion rate of carbon dioxide to | <p class="pcontent">To measure the enzyme activity of CA, we compare the conversion rate of carbon dioxide to | ||
bicarbonate ion. After saturated CO<sub>2</sub> solution is prepared, we add fixed amount of bacteria broth that | bicarbonate ion. After saturated CO<sub>2</sub> solution is prepared, we add fixed amount of bacteria broth that | ||
| − | contains CA construction into the solution. We then measure the time | + | contains CA construction into the solution. We then measure the time taken for the pH value to decrease from 8.3 to 6.3. |
We compare the measured time interval with the time interval that enzyme was not added to determine the enzyme activity of CA. | We compare the measured time interval with the time interval that enzyme was not added to determine the enzyme activity of CA. | ||
</p> | </p> | ||
| Line 176: | Line 176: | ||
Previously, every basic part was the backbone conserved in the backbone of pSB1C3. | Previously, every basic part was the backbone conserved in the backbone of pSB1C3. | ||
We then link the construction together and | We then link the construction together and | ||
| − | even change the backbone of some composite parts to pSB3K3 for lower protein expression. | + | even change the backbone of some composite parts to pSB3K3 for a lower protein expression. |
</p> | </p> | ||
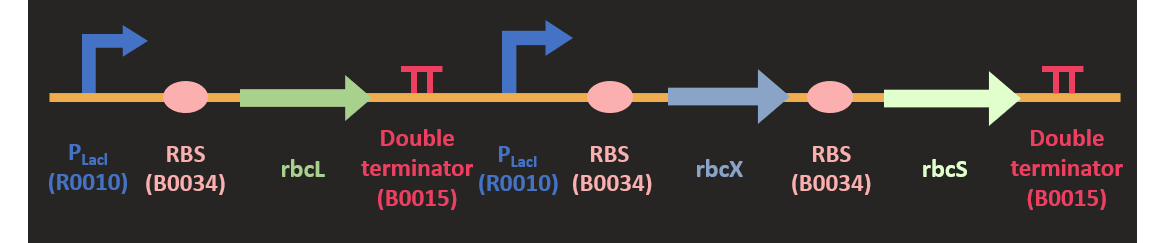
<h5 class="question">Rubisco whole protein in pSB1C3</h5> | <h5 class="question">Rubisco whole protein in pSB1C3</h5> | ||
| − | <p class="pcontent">We link each basic | + | <p class="pcontent">We link each basic part together with biobrick standard method. |
We link P<sub>T7</sub>-<i>rbcL</i> and P<sub>T7</sub>-<i>rbcX</i>-<i>rbcS</i> together. | We link P<sub>T7</sub>-<i>rbcL</i> and P<sub>T7</sub>-<i>rbcX</i>-<i>rbcS</i> together. | ||
| − | The former, the insert, was digested with EcoRI and SpeI and the | + | The former, the insert, was digested with EcoRI and SpeI and the latter, the backbone, |
is digested with EcoRI and XbaI. | is digested with EcoRI and XbaI. | ||
We ligate the backbone with the insert to complete this composite part. | We ligate the backbone with the insert to complete this composite part. | ||
</p> | </p> | ||
<h5 class="question"><i>prk</i> gene into pSB3K3</h5> | <h5 class="question"><i>prk</i> gene into pSB3K3</h5> | ||
| − | <p class="pcontent">PRK catalyzes the reaction of | + | <p class="pcontent">PRK catalyzes the reaction of converting Ru5P into RuBP. |
Not native to the host, RuBP is, nonetheless, toxic to <i>E. coli</i>. | Not native to the host, RuBP is, nonetheless, toxic to <i>E. coli</i>. | ||
| − | We hope that expression of PRK | + | We hope that the expression of PRK could be lower in the host so we change the backbone of it into pSB3K3. |
We selected J04450 from the distributed kit that under the backbone of pSB3K3, | We selected J04450 from the distributed kit that under the backbone of pSB3K3, | ||
which will express red color after the formation of the colony. We digest both backbone and insert with EcoRI and PstI and ligate both fragments. | which will express red color after the formation of the colony. We digest both backbone and insert with EcoRI and PstI and ligate both fragments. | ||
Latest revision as of 15:18, 3 November 2018