Oscarliu117 (Talk | contribs) |
Oscarliu117 (Talk | contribs) |
||
| Line 63: | Line 63: | ||
<ul> | <ul> | ||
| − | <li>Experiment 1:Instrument calibration</li> | + | <li>Experiment 1:Instrument calibration</li></br> |
<ol> | <ol> | ||
| − | <li>Experimental purpose</li> | + | <li>Experimental purpose</li></br> |
<ul> | <ul> | ||
<li>Because the buffer solution wouldn’t change too much in the | <li>Because the buffer solution wouldn’t change too much in the | ||
environment, we used the buffer solution to calibrate the pH meter to | environment, we used the buffer solution to calibrate the pH meter to | ||
| − | ensure that the deviation would the same every time.</li> | + | ensure that the deviation would the same every time.</li></br> |
</ul> | </ul> | ||
| − | <li>Experimental method</li> | + | <li>Experimental method</li></br> |
<ol type="a"> | <ol type="a"> | ||
<li>Insert pH meter into pH 7 buffer solution, wait about 1 min, it | <li>Insert pH meter into pH 7 buffer solution, wait about 1 min, it | ||
will achieve a stable | will achieve a stable | ||
| − | value.</li> | + | value.</li></br> |
<li>Minus the value with pH 7, and it will get the offset value. For | <li>Minus the value with pH 7, and it will get the offset value. For | ||
| − | instance, 7-7.09=-0.09.</li> | + | instance, 7-7.09=-0.09.</li></br> |
<div id="centerimg" class="pic1"> | <div id="centerimg" class="pic1"> | ||
| Line 87: | Line 87: | ||
</div> | </div> | ||
| + | </br> | ||
<li>Write the offset value into code, upload the code into Arduino | <li>Write the offset value into code, upload the code into Arduino | ||
| − | again.</li> | + | again.</li></br> |
<div id="centerimg" class="pic2"> | <div id="centerimg" class="pic2"> | ||
| Line 94: | Line 95: | ||
</div> | </div> | ||
| + | </br> | ||
<li>After rinsing the pH meter, insert it into pH 4 buffer | <li>After rinsing the pH meter, insert it into pH 4 buffer | ||
| − | solution.</li> | + | solution.</li></br> |
<li>If it is found to be too different from the error of pH=4, you | <li>If it is found to be too different from the error of pH=4, you | ||
should adjust the knob. | should adjust the knob. | ||
After inserting pH = 4, it is found that the error with 4 is very | After inserting pH = 4, it is found that the error with 4 is very | ||
| − | large.</li> | + | large.</li></br> |
<div id="centerimg" class="pic3"> | <div id="centerimg" class="pic3"> | ||
| Line 105: | Line 107: | ||
</div> | </div> | ||
| + | </br> | ||
<li>Adjust the knob. (Remark: Because the change of the knob is small, | <li>Adjust the knob. (Remark: Because the change of the knob is small, | ||
it may have to turn a | it may have to turn a | ||
| − | few more times until display pH 4.)</li> | + | few more times until display pH 4.)</li></br> |
<div id="centerimg" class="pic4"> | <div id="centerimg" class="pic4"> | ||
<img class="oneimg" src=""> | <img class="oneimg" src=""> | ||
</div> | </div> | ||
| − | + | </br> | |
| − | <li>Finally, it can be found that the measured pH is 4.</li> | + | <li>Finally, it can be found that the measured pH is 4.</li></br> |
<div id="centerimg" class="pic5"> | <div id="centerimg" class="pic5"> | ||
| Line 122: | Line 125: | ||
</ol> | </ol> | ||
| − | + | </br> | |
| − | <li>Formula calculation</li> | + | <li>Formula calculation</li></br> |
<ol type="a"> | <ol type="a"> | ||
| − | <li>voltage = analog value*5/1024</li> | + | <li>voltage = analog value*5/1024</li></br> |
| − | <li>pH value = 3.5*voltage+offset</ | + | <li>pH value = 3.5*voltage+offset</li></br> |
</ol> | </ol> | ||
| − | <li>Experiment 2: Precision measurement</li> | + | <li>Experiment 2: Precision measurement</li></br> |
<ol> | <ol> | ||
| − | <li>Purpose</li> | + | <li>Purpose</li></br> |
<ul> | <ul> | ||
<li>We assume that pH 4 to 7 is linear, so we want to verify | <li>We assume that pH 4 to 7 is linear, so we want to verify | ||
whether it is linear between pH | whether it is linear between pH | ||
4 to 7. By measuring the solution of pH 4.7, compare its | 4 to 7. By measuring the solution of pH 4.7, compare its | ||
| − | deviation.</li> | + | deviation.</li></br> |
</ul> | </ul> | ||
| − | <li>Method</li> | + | <li>Method</li></br> |
<ul> | <ul> | ||
<li>Mixing a solution with pH=4.7 and measure with a calibrated pH | <li>Mixing a solution with pH=4.7 and measure with a calibrated pH | ||
| − | meter.</li> | + | meter.</li></br> |
</ul> | </ul> | ||
| − | <li>Result</li> | + | <li>Result</li></br> |
<ul> | <ul> | ||
| Line 157: | Line 160: | ||
value was stable at pH 4.83 | value was stable at pH 4.83 | ||
with an error of about 0.13, which roughly met the error of | with an error of about 0.13, which roughly met the error of | ||
| − | this pH meter ± 0.1 pH.</li> | + | this pH meter ± 0.1 pH.</li></br> |
</ul> | </ul> | ||
| Line 164: | Line 167: | ||
</div> | </div> | ||
| − | + | </br> | |
| + | <li>Materials used</li></br> | ||
<ol> | <ol> | ||
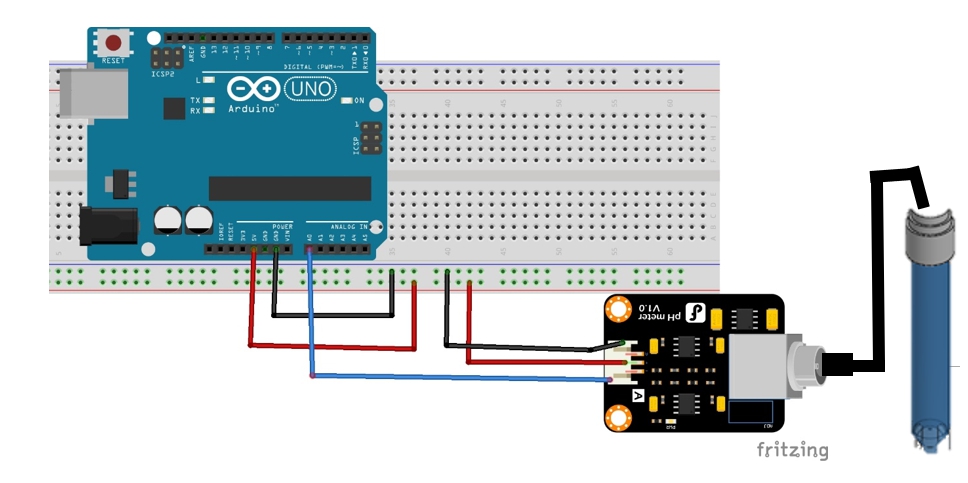
| − | <li>Arduino UNO</li> | + | <li>Arduino UNO</li></br> |
| − | <li>pH sensor | + | <li>pH sensor</li></br> |
<ol type="a"> | <ol type="a"> | ||
<li>Module Power : 5.00V</li> | <li>Module Power : 5.00V</li> | ||
| Line 175: | Line 179: | ||
<li>Accuracy : ± 0.1pH (25 ℃)</li> | <li>Accuracy : ± 0.1pH (25 ℃)</li> | ||
</ol> | </ol> | ||
| − | </ | + | </br> |
| − | <li>pH buffer solution</li> | + | <li>pH buffer solution</li></br> |
| − | <li>Arduino code</li> | + | <li>Arduino code</li></br> |
</ol> | </ol> | ||
Revision as of 12:49, 30 September 2018
pH Meter