

Description
Amyotrophic lateral sclerosis (ALS), also known as Lou Gehrig’s disease, is a devastating neurodegenerative disease characterized by the progressive degeneration of motor neurons in the brain and spinal cord1. The disease is fatal, with an average survival rate of only 3–5 years after diagnosis2. The exact cause of ALS is still unknown. Several genetic factors have been identified and are currently being investigated further. No specific environmental factor has been definitively shown to cause ALS3. ALS can be classified as familial or sporadic, depending on whether or not there is a family history of the disease. Familial ALS (FALS) accounts for 10% of all ALS cases, while Sporadic ALS (SALS) accounts for the other 90%4 Although SALS and FALS appear identical clinically and pathologically, the genetics of FALS are better understood. Therefore, the medications developed thus far are mainly based on the research conducted on FALS rather than SALS.
A.L.S Pathophysiology – Reactive Gliosis
The defining feature of ALS is the death of motor neurons in the motor cortex, brainstem and the spinal cord2. It is not fully understood why neurons die in ALS, but this neurodegeneration is thought to involve many different cellular and molecular processes5.
Recent evidence suggests that in both SALS and FALS, non-neuronal cells, astrocytes and microglia, directly contribute to motor neuronal damage and cell death. Under disease or injury conditions, astrocytes change their normal morphology and function, gaining a new neurotoxic function- rapidly killing motor neurons6-7. These so called "reactive astrocytes" change their gene expression relative to quiescent astrocytes. Two such distinguishing genetic markers are Steap4 and Timp1 genes, expressed exclusively in reactive astrocytes8-11.

A recent study has shown that activated microglia cells are required to induce reactive astrocytes, by secreting cytokines such as IL-1α and TNF-α12. The synthesis of both these cytokines is mediated by the NF-kB transcription factor13. NF-kB activation in microglia causes motor neurons death in vitro as well as in vivo. Heterozygous inhibition of NF-kB in microglia substantially delayed disease progression in ALS mice model14.
The process where microglia secrete an excess of cytokines and induce reactive astrocytes is an inflammatory process called reactive gliosis. Reactive gliosis has been identified as a common process to ALS and other neurodegenerative diseases, such as Huntington’s and Parkinson’s disease12.
Motivation
To date, there is no cure for ALS. Available medications focus on treating symptoms and providing supportive care, with the goal of improving quality of life and prolonging patient survival5. Currently there are two main medications sold in the market for treating ALS: Riluzole and Edaravone.
Riluzole has been found to modestly prolong survival by about 2-3 months15, although the mechanism of its action is poorly understood. Edaravone has been shown to slow the decline in only a small group of patients who meet very specific criteria16.
To date, there is no evidence that either Riluzole or Edaravone are significantly effective in prolonging survival in most cases of ALS (FALS and SALS alike)17.
OriginALS Goal
Our objective, as the BGU-IGEM team, is to develop a therapeutic system that will ultimately prolong survival of ALS patients via novel genetic engineering techniques.
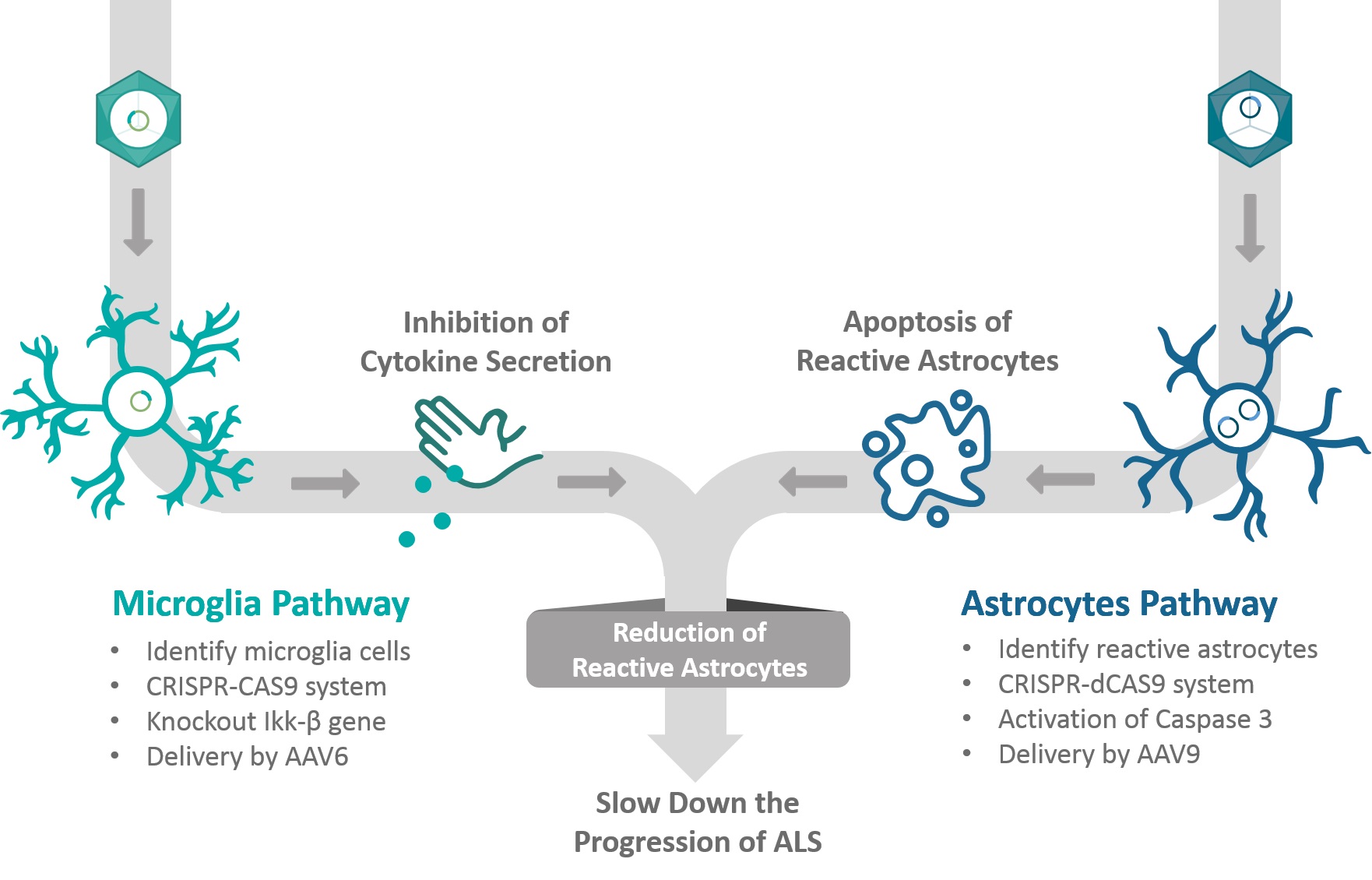
A Two Dimensional Approach
We divide our project into two approaches: 1) identify and specifically target reactive astrocytes (and not normal resting astrocytes) by their distinguishing markers and prevent their toxic activities by promoting an intrinsic apoptotic signal 2) inhibit the secretion of toxic pro-inflammatory cytokines by microglia cells.
The Astrocyte Pathway:
In the astrocyte pathway, we aim to identify and eliminate only reactive astrocytes. In order to identify the reactive astrocytes, we will use the promoters of the distinguishing markers, Steap4 and Timp1. The Timp1 and Steap4 genes have high levels of expression specifically in reactive astrocyte when compared to other cells in the central nervous system (CNS)18.
These two promoters will control the expression of dCas9 and its accompanying gRNA19. Each component in our CRISPR system is placed on a different plasmid and have a different promoter: the dCas9 protein and the transcription factors will be expressed under the pTimp1 promoter, while the gRNA will be expressed under the pSteap4 promoter, with ribozyme flanked RNA.

Only if both plasmids will enter the target cell and these promoters will be activated, the dCas9 and the gRNA will join together, to target a synthetic promoter that express an exogenous Caspase3.
In order to add further specificity, we plan to deliver these plasmids using the Adeno-Associated Virus-9 (AAV9)20. This modified virus targets and delivers its contents specifically to astrocyte cells21.
The activation of caspase 3 by the dCas9 system will eventually lead to apoptotic death of the reactive astrocytes22.
The Microglia Pathway:
In the microglia pathway, we will knockout the IKKβ gene in microglia cells, using the CRISPR-Cas9 system23 delivered by AAV620 virus that targets microglia cells24. The IKKβ gene is transcribed and translated into a kinase which has a role as an activator upstream to the NFkB complex assembly. Therefore, the knockout will ensure a decrease in the neuro-inflammation transducing classical NFkB signal25.

Aiming for specific microglia expression, we chose to combine the pF4/80 promoter, which is a known macrophage marker26 with AAV6 viral vector. The pF4/80 promoter will be responsible for the expression of the Cas9 protein, while the U6 promoter, which is a non-specific promoter, is responsible for the expression of the IKKβ guide RNA sequence.
Our CRISPR Cas9 system will target and knock out the IKKβ gene, resulting in inhibition of inflammatory cytokines synthesis and thus reduce the formation of new reactive astrocytes in the system.
Summary
To summarize, in our two-dimensional approach, we will target reactive astrocytes and microglia cells.
The goal of the astrocyte pathway is to remove the harmful activity of existing reactive astrocytes. This will be achieved by inducing apoptosis in the existing reactive astrocytes. All this without damaging the normal resting astrocytes.
In the microglia pathway we want to prevent the formation of new reactive astrocytes. We plan to achieve our goal by reducing the secretion of cytokines from activated microglia.
This novel combined approach is aimed to break the cycle of unregulated reactive gliosis, and hopefully will slow down the progression rate of both familial and sporadic ALS. By combining the removal of toxic cells with the prevention of the formation of new ones, the number of toxic astrocytes can be significantly reduced, thus slowing down the progression rate of ALS.
Future Applications

References
- Roche, Jose C., et al. "A proposed staging system for amyotrophic lateral sclerosis." Brain3 (2012): 847-852.
- Robberecht, Wim, and Thomas Philips. "The changing scene of amyotrophic lateral sclerosis." Nature Reviews Neuroscience4 (2013): 248.
- Al-Chalabi, Ammar, and Orla Hardiman. "The epidemiology of ALS: a conspiracy of genes, environment and time." Nature Reviews Neurology11 (2013): 617.
- Mulder, Donald W., et al. "Familial adult motor neuron disease: amyotrophic lateral sclerosis." Neurology4 (1986): 511-517.
- van Es, Michael A., et al. "Amyotrophic lateral sclerosis." The Lancet(2017).
- Lee, Junghee, et al. "Astrocytes and microglia as non-cell autonomous players in the pathogenesis of ALS." Experimental neurobiology5 (2016): 233-240.
- Tripathi, Pratibha, et al. "Reactive astrocytes promote ALS-like degeneration and intracellular protein aggregation in human motor neurons by disrupting autophagy through TGF-β1." Stem cell reports2 (2017): 667-680.
- Zamanian, Jennifer L., et al. "Genomic analysis of reactive astrogliosis." Journal of neuroscience18 (2012): 6391-6410.
- Zhang, Ye, et al. "An RNA-sequencing transcriptome and splicing database of glia, neurons, and vascular cells of the cerebral cortex." Journal of Neuroscience36 (2014): 11929-11947.
- Tokuda, Eiichi, Eriko Okawa, and Shin‐ichi Ono. "Dysregulation of intracellular copper trafficking pathway in a mouse model of mutant copper/zinc superoxide dismutase‐linked familial amyotrophic lateral sclerosis." Journal of neurochemistry1 (2009): 181-191.
- Lorenzl, S., et al. "Tissue inhibitors of matrix metalloproteinases are elevated in cerebrospinal fluid of neurodegenerative diseases." Journal of the neurological sciences1-2 (2003): 71-76.
- Liddelow, Shane A., et al. "Neurotoxic reactive astrocytes are induced by activated microglia." Nature7638 (2017): 481.
- Tak, Paul P., and Gary S. Firestein. "NF-κB: a key role in inflammatory diseases." The Journal of clinical investigation1 (2001): 7-11.
- Frakes, Ashley E., et al. "Microglia induce motor neuron death via the classical NF-κB pathway in amyotrophic lateral sclerosis." Neuron5 (2014): 1009-1023.
- Miller, Robert G., JD and Mitchell, and Dan H. Moore. "Riluzole for amyotrophic lateral sclerosis (ALS)/motor neuron disease (MND)." Cochrane database of systematic reviews3 (2012).
- Abe, Koji, et al. "Safety and efficacy of edaravone in well defined patients with amyotrophic lateral sclerosis: a randomised, double-blind, placebo-controlled trial." The Lancet Neurology7 (2017): 505-512.
- Petrov, Dmitry, et al. "ALS clinical trials review: 20 years of failure. Are we any closer to registering a new treatment?." Frontiers in aging neuroscience9 (2017): 68.
- Trakhtenberg, Ephraim F., et al. "Cell types differ in global coordination of splicing and proportion of highly expressed genes." Scientific Reports6 (2016): 32249.
- Perez-Pinera, Pablo, et al. "RNA-guided gene activation by CRISPR-Cas9–based transcription factors." Nature methods10 (2013): 973.
- Daya, Shyam, and Kenneth I. Berns. "Gene therapy using adeno-associated virus vectors." Clinical microbiology reviews4 (2008): 583-593.
- Foust, Kevin D., et al. "Intravascular AAV9 preferentially targets neonatal neurons and adult astrocytes." Nature biotechnology1 (2008): 59.
- Srinivasula, Srinivasa M., et al. "Generation of constitutively active recombinant caspases-3 and-6 by rearrangement of their subunits." Journal of Biological Chemistry17 (1998): 10107-10111.
- Cong, Le, and Feng Zhang. "Genome engineering using CRISPR-Cas9 system." Chromosomal Mutagenesis. Humana Press, New York, NY, 2015. 197-217.
- Rosario, Awilda M., et al. "Microglia-specific targeting by novel capsid-modified AAV6 vectors." Molecular Therapy-Methods & Clinical Development3 (2016).
- Frakes, Ashley E., et al. "Microglia induce motor neuron death via the classical NF-κB pathway in amyotrophic lateral sclerosis." Neuron5 (2014): 1009-1023.
- Austyn, Jonathan M., and Siamon Gordon. "F4/80, a monoclonal antibody directed specifically against the mouse macrophage." European journal of immunology10 (1981): 805-815.

