| Line 306: | Line 306: | ||
<p class="F2"> | <p class="F2"> | ||
<img src="https://static.igem.org/mediawiki/2018/c/ce/T--XMU-China--OMVs22.png"> | <img src="https://static.igem.org/mediawiki/2018/c/ce/T--XMU-China--OMVs22.png"> | ||
| − | <p class="Figure_word">Figure 2. a) Genetic circuit of SpyTag-OmpA part and OmpA-SpyTag part. b) Schematic illustration of module one.</p> | + | <p class="Figure_word"><strong>Figure 2.</strong> a) Genetic circuit of SpyTag-OmpA part and OmpA-SpyTag part. b) Schematic illustration of module one.</p> |
</p> | </p> | ||
<p>Furthermore, to characterize whether our OmpA-SpyTag can localize in OMVs, we also attach GFP to both OmpA parts (Figure 3). </p> | <p>Furthermore, to characterize whether our OmpA-SpyTag can localize in OMVs, we also attach GFP to both OmpA parts (Figure 3). </p> | ||
<p class="F2"> | <p class="F2"> | ||
<img src="https://static.igem.org/mediawiki/2018/d/db/T--XMU-China--OMVs23.png"> | <img src="https://static.igem.org/mediawiki/2018/d/db/T--XMU-China--OMVs23.png"> | ||
| − | <p class="Figure_word">Figure 3. a) Genetic circuit of SpyTag-OmpA-GFP part and OmpA-SpyTag-GFP part. b) Schematic illustration of these two parts.</p> | + | <p class="Figure_word"><strong>Figure 3.</strong> a) Genetic circuit of SpyTag-OmpA-GFP part and OmpA-SpyTag-GFP part. b) Schematic illustration of these two parts.</p> |
</p> | </p> | ||
<h1>Module two – L7Ae</h1> | <h1>Module two – L7Ae</h1> | ||
<p>Archaeal ribosomal protein protein L7Ae owns the ability to bind with RNA with C/Dbox RNA structure.<sup>[2]-[4]</sup> Through the isopeptide bonds formed between SpyTag and SpyCatcher, L7Ae can be anchored with OmpA, and then secreted into OMVs. To transport our L7Ae into the periplasmic space, we insert TorA leader sequence to the circuit (Figure 4). </p> | <p>Archaeal ribosomal protein protein L7Ae owns the ability to bind with RNA with C/Dbox RNA structure.<sup>[2]-[4]</sup> Through the isopeptide bonds formed between SpyTag and SpyCatcher, L7Ae can be anchored with OmpA, and then secreted into OMVs. To transport our L7Ae into the periplasmic space, we insert TorA leader sequence to the circuit (Figure 4). </p> | ||
<p class="F2"><img src="https://static.igem.org/mediawiki/2018/c/c3/T--XMU-China--OMVs24.png"> | <p class="F2"><img src="https://static.igem.org/mediawiki/2018/c/c3/T--XMU-China--OMVs24.png"> | ||
| − | <p class="Figure_word">Figure 4. Genetic circuit of SpyCatcher-L7Ae.</p> | + | <p class="Figure_word"><strong>Figure 4.</strong> Genetic circuit of SpyCatcher-L7Ae.</p> |
</p> | </p> | ||
<p>To localize our L7Ae inside OMVs, we also conjugate mRFP1 to the L7Ae-SpyCatcher. Under the induction of the IPTG, our bacteria will express our L7Ae parts (Figure 5). </p> | <p>To localize our L7Ae inside OMVs, we also conjugate mRFP1 to the L7Ae-SpyCatcher. Under the induction of the IPTG, our bacteria will express our L7Ae parts (Figure 5). </p> | ||
<p class="F2"><img src="https://static.igem.org/mediawiki/2018/3/37/T--XMU-China--OMVs25.png"> | <p class="F2"><img src="https://static.igem.org/mediawiki/2018/3/37/T--XMU-China--OMVs25.png"> | ||
| − | <p class="Figure_word">Figure 5. Genetic circuit of SpyCatcher-L7Ae-RFP.</p> | + | <p class="Figure_word"><strong>Figure 5.</strong> Genetic circuit of SpyCatcher-L7Ae-RFP.</p> |
</p> | </p> | ||
<h1>Module three - Box</h1> | <h1>Module three - Box</h1> | ||
| Line 329: | Line 329: | ||
<p>Under the induction of 0.2% Arabinose and then 0.5 mM IPTG, we can produce L7Ae-SpyCatcher and siRNA-C/D<sub>box</sub>. With the isopeptide bond formed between SpyTag and SpyCatcher, and the ability of L7Ae to be bind with C/D<sub>box</sub>, we can produce customizable and cell-free OMVs containing specific siRNA to target for oncogenic gene (Figure 7). </p> | <p>Under the induction of 0.2% Arabinose and then 0.5 mM IPTG, we can produce L7Ae-SpyCatcher and siRNA-C/D<sub>box</sub>. With the isopeptide bond formed between SpyTag and SpyCatcher, and the ability of L7Ae to be bind with C/D<sub>box</sub>, we can produce customizable and cell-free OMVs containing specific siRNA to target for oncogenic gene (Figure 7). </p> | ||
<p class="F2"><img src="https://static.igem.org/mediawiki/2018/a/ac/T--XMU-China--OMVs27.png"> | <p class="F2"><img src="https://static.igem.org/mediawiki/2018/a/ac/T--XMU-China--OMVs27.png"> | ||
| − | <p class="Figure_word">Figure 6. Genetic circuit of Box part.</p> | + | <p class="Figure_word"><strong>Figure 6.</strong> Genetic circuit of Box part.</p> |
</p> | </p> | ||
<h1 class="references">References</h1> | <h1 class="references">References</h1> | ||
Revision as of 14:34, 17 October 2018


Ideas
During our brainstorming, we read the project of the 2017 Munich team, which established a system for the universal detection of RNA biomarkers. Therefore, we hope to improve the entire detection system and establish a system that can detect protein biomarkers universally.
Luckily, our ABCD system has appeared. Later, we discussed how could it be implied in synthetic biology. We found the answer ultimately in cell-free synthetic biology. In addition, after exchanging ideas with the Munich team, we were more certain that we did use engineering principles to re-edit the biological system.
We have divided our ABCD system into three modules, using engineering ideas, so that each can be "plug and play", but also can be "replaced". The three modules are "Sensor", "Amplifier" and "Reporter".

Sensor
In the Sensor, we first selected the epithelial cell adhesion molecule as the object of our detection. The epithelial cell adhesion molecule (EpCAM, also known as ESA or CD326) is a transmembrane glycoprotein that mediates epithelial cell-specific intercellular adhesion and involves cell signaling, migration, proliferation, and differentiation. EpCAM has been detected in most adenocarcinomas, and is also found in metastatic tumors, malignant effusions and cancer stem cells.
Next, we chose the ssDNA aptamer as an intermediary for downstream signaling of proteins. It is a single-stranded DNA that specifically binds to a target molecule. Theoretically, the corresponding aptamer of any protein can be screened by SELEX technique. And we choose EpCAM's aptamer-SYL3C.[1]
| Name | Sequence | Modification |
|---|---|---|
| SYL3C | 5'-CACTACAGAGGTTGCGTCTGTCCC ACGTTGTCATGGGGGGTTGGCCTG-3' | 3'-Biotin-TEG |
| Complementary strand | 5'-CTCTGTAGTGTTTTTTTTTTTTTT-3' | 3'-FITC |
At the same time, we also use the competition method to facilitate our design of hardware. As shown in the figure, according to our assumption, the upstream EpCAM will compete with our designed aptamer and its partial complementary chain. After that, some complementary chains will be competed and freed, coming out and going to the next module to get the signal amplified.

For this module, we also modeled the molecular docking model of aptamer and protein, and partially predicted the number of bases in the complementary strands. Click to learn more.
Amplifier
TIn this module we used the Cas12a protein to amplify the signal. Cas12a (also known as Cpf1) was originally discovered as a replacement for Cas9.[2] But this year's discovery showed that it had a new function of being activated by ssDNA and realizing trans-cleavage.[3]

As shown in the figure, when some of our upstream complementary strands are released to the Cas12a protein after competitions, recognized by the specifically designed crRNA and finally complemented with it successfully, the Cas12a protein will be activated, and non-specific digestion will occur.
| Name | Sequence | Modification |
|---|---|---|
| SYL3C-crRNA | 5'-UAAUUUCUACUCUUGUAGAU AAAAAAAAAAACACUACAGAG-3 | None |
Reporter
When the Cas12a protein is activated, it will realize a non-specific cleavage that functions as DNase I, so we can report our signal by using the method of detecting DNase I.
We finally used the fluorescent signal as our reporting signal, because it was easy to observe, semi-quantitative, and can be directly read and analyzed by the machine. And our signal reports used the commercially available DNaseAlert kit on the market.[4]
The working principle is as follows: a combination of a ground state fluorescent molecule and a quencher generates a complex by ssDNA, and the complex quenches the fluorescence. When the nuclease digests the ssDNA, the quenching fails and the fluorophore re-emits.

References
[1] Yanling Song, Zhi Zhu, Yuan An, Weiting Zhang, Huimin Zhang, Dan Liu, Chundong Yu, Wei Duan, Chaoyong James Yang, Selection of DNA Aptamers against Epithelial Cell Adhesion Molecule for Cancer Cell Imaging and Circulating Tumor Cell Capture. Anal Chem. 2013, 85: 4141-4149.
[2] Zetsche B, Gootenberg JS, Abudayyeh OO, Slaymaker IM, Makarova KS, Essletzbichler P, Volz SE, Joung J, van der Oost J, Regev A, Koonin EV, Zhang F, Cpf1 is a single RNA-guided endonuclease of a class 2 CRISPR-Cas system. Cell. 2015, 163(3): 759-771.
[3] Chen JS, Ma E, Harrington LB, Da Costa M, Tian X, Palefsky JM, Doudna JA, CRISPR-Cas12a target binding unleashes indiscriminate single-stranded DNase activity. Science. 2018, 360(6387): eaar6245.
[4] DNaseAlert™ Substrate Nuclease Detection System User Manual.
Ideas
To utilize outer-membrane vesicles (OMVs) as our cell-free platform for RNA delivery and then for disease treatment, we design three modules with different functions, which can be divided into OmpA, L7Ae, and Box (Figure 1).

Figure 1. a) Genetic circuit of three modules. b) Schematic illustration of our total circuit.
Module one - OmpA
Porin outer-membrane protein A, OmpA, is a kind of highly expressed transmembrane porin protein of E. coli and is therefore abundant in OMVs.[1] We insert SpyTag to the C-termini and N-termini of a truncated form of OmpA respectively to compare the efficiency of SpyTag/SpyCatcher linkage (Figure 2).

Figure 2. a) Genetic circuit of SpyTag-OmpA part and OmpA-SpyTag part. b) Schematic illustration of module one.
Furthermore, to characterize whether our OmpA-SpyTag can localize in OMVs, we also attach GFP to both OmpA parts (Figure 3).

Figure 3. a) Genetic circuit of SpyTag-OmpA-GFP part and OmpA-SpyTag-GFP part. b) Schematic illustration of these two parts.
Module two – L7Ae
Archaeal ribosomal protein protein L7Ae owns the ability to bind with RNA with C/Dbox RNA structure.[2]-[4] Through the isopeptide bonds formed between SpyTag and SpyCatcher, L7Ae can be anchored with OmpA, and then secreted into OMVs. To transport our L7Ae into the periplasmic space, we insert TorA leader sequence to the circuit (Figure 4).

Figure 4. Genetic circuit of SpyCatcher-L7Ae.
To localize our L7Ae inside OMVs, we also conjugate mRFP1 to the L7Ae-SpyCatcher. Under the induction of the IPTG, our bacteria will express our L7Ae parts (Figure 5).

Figure 5. Genetic circuit of SpyCatcher-L7Ae-RFP.
Module three - Box
We insert a C/Dbox into the 3'-untranslated region (3'-UTR) of the gene coding for siRNA.[5] Under the induction of 0.2% Arabinose, our bacteria will express our Box parts (Figure 6).

Figure 6. Genetic circuit of Box part.
Under the induction of 0.2% Arabinose and then 0.5 mM IPTG, we can produce L7Ae-SpyCatcher and siRNA-C/Dbox. With the isopeptide bond formed between SpyTag and SpyCatcher, and the ability of L7Ae to be bind with C/Dbox, we can produce customizable and cell-free OMVs containing specific siRNA to target for oncogenic gene (Figure 7).
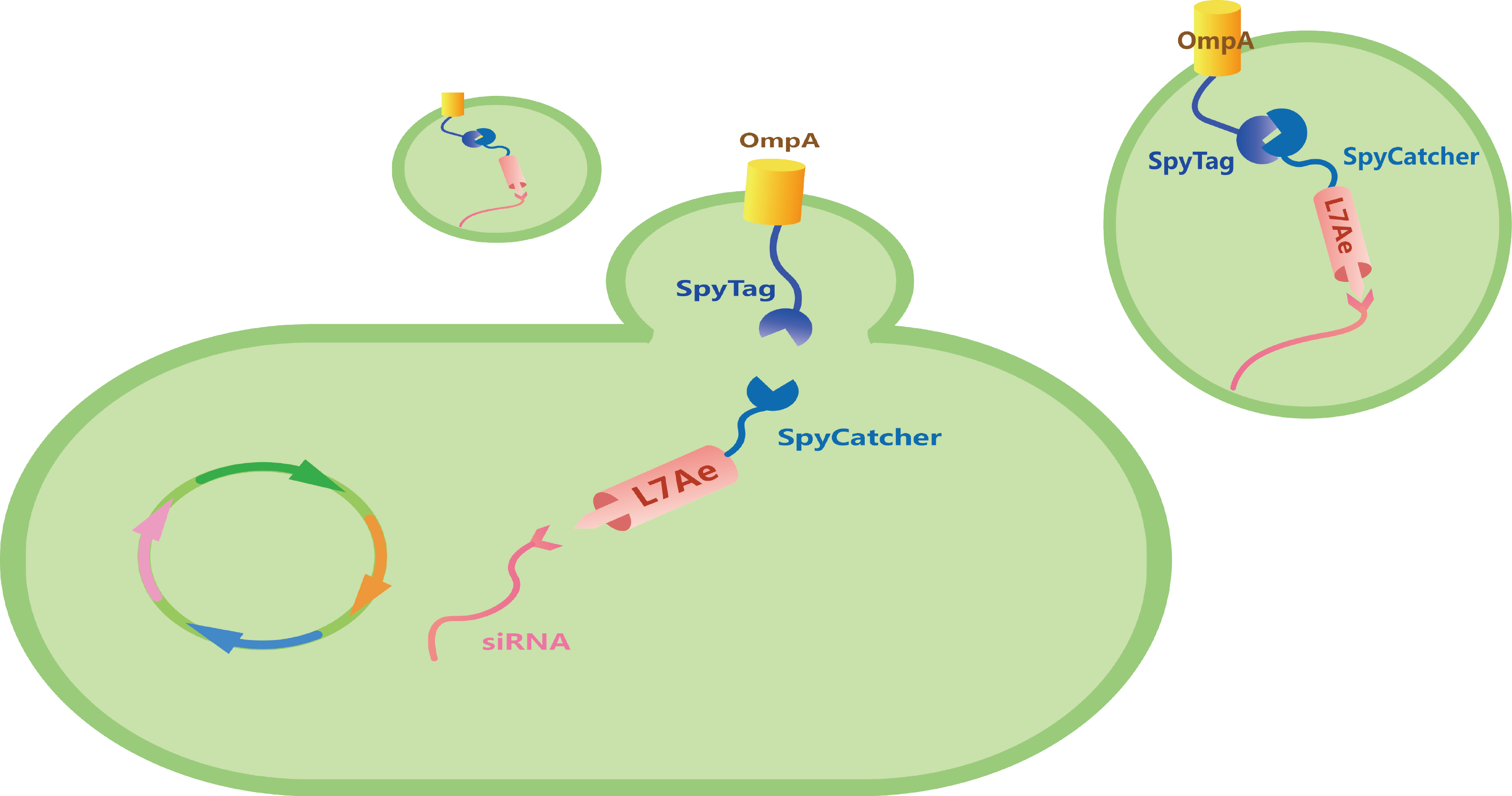
Figure 6. Genetic circuit of Box part.
References
[1] Acevedo R, Fernández S, Zayas C, et al. Bacterial Outer Membrane Vesicles and Vaccine Applications. Frontiers in Immunology. 2014, 5: 121.
[2] Auslã¤Nder S, Auslã¤Nder D, Mã¼Ller M, et al. Programmable single-cell mammalian biocomputers. Nature. 2012, 487(7405): 123-127.
[3] Saito H, Fujita Y, Kashida S, et al. Synthetic human cell fate regulation by protein-driven RNA switches. Nature Communications. 2011, 2(1): 160.
[4] Saito H, Kobayashi T, Hara T, et al. Synthetic translational regulation by an L7Ae–kink-turn RNP switch. Nature Chemical Biology. 2009, 6(1): 71.
[5] Kojima R, Bojar D, Rizzi G, et al. Designer exosomes produced by implanted cells intracerebrally deliver therapeutic cargo for Parkinson's disease treatment. Nature Communications. 2018, 9(1): 1305.
Ideas
We selected two proteins SAHS1 and CAHS1, which had shown excellent effects worked out by the Japanese team in 2012 years. SAHS1 was used to preserve the protein and CAHS1 was used for the preservation of the bacteria.
Following SAHS1, a his-tag is connected via a linker to facilitate protein purification. A blue chromoprotein is also attached to the circuit as a reporter gene. After transformation into E.coli BL21 for expression, the purified TDP proteins are dealt with different concentrations of TDP proteins, and then the proteins are lyophilized and dried, based on what we need to design for the control group to verify the biological function of SAHS protein. As a result, a kit for storing proteins more conveniently has been developed, or SAHS protein has been used to simplify existing methods for preserving proteins.
Here are the gene circuits we designed:

Figure1. Our gene circuits.
CAHS protein is a cytoplasmic-abundant protein which can strength the bacteria stress tolerance. So taking biosafety into account, we add a constant promoter into the CAHS circuit to express Lac I in order to inhabit the initiation of the Lac O promoter which can only be activated and then expressing CAHS protein and mRFP after IPTG is added. And the mRFP is added for homologous recombination into the genome of the bacteria for screening. We intend to adopt the Red recombination method to transform the circuit into the genome of DH5α, thereby improving DH5α and making DH5α easier to be preserved.
We decided to add a mRFP or blue chromoprotein which follows the SAHS gene to help us select the E.coli that contains the designed circuit by color. At the same time, since the SAHS protein is an exogenous protein, it is preceded by a signal peptide, which allows it to pass through the cell membrane. Therefore, we designed to obtain our target protein of high concentration by centrifuging the supernatant, removing the LB medium, and finally performing the protein concentration. As a protective agent, SAHS can prevent Cas12a from lyophilization.
Expectation
A new E.coli strain
We intend to transform the circuit into the genome of DH5α, thereby improving DH5α and making DH5α easier to be preserved. And by this way, we can save and transport the DH5α normally and easier.
A new way to preserve protein
We hope that the purified TDPs can help preserve the proteins and other biological materials. And we also want to produce a kit for preserve these biological materials.
References
[1] Yamaguchi A. Two Novel Heat-Soluble Protein Families Abundantly Expressed in an Anhydrobiotic Tardigrade. PLoS ONE. 2012, 7(8): e44209.
[2] Boothby TC. Tardigrades Use Intrinsically Disordered Proteins to Survive Desiccation. Mol Cell. 2017 Mar16, 65(6): 975-984.e5.
[3] Boothby TC. Intrinsically Disordered Proteins and Desiccation Tolerance: Elucidating Functional and Mechanistic Underpinnings of Anhydrobiosis. Bioessays. 2017 Nov, 39(11): 1700119.

