Contents
Proof of Concept
Glycolic acid production in E. coli
Our aim was to produce glycolic acid for the production of biodegradable polymers. We were able to overexpress the genes aceA and ycdW in Escherichia coli. The proteins AceA and YcdW were his-tagged and purified via an ÄKTA system. The purification was confirmed by an SDS-PAGE and a western blot. After that, we successfully characterized YcdW with an NADPH dependent enzyme assay. The assay indicates that YcdW uses glyoxylate as substrate under consumption of NADPH to produce glycolic acid. The functionality of AceA was verified with a phenylhydrazine-dependent assay. AceA catalyzes the reaction from isocitrate to glyoxylate. The enzyme assays were performed a second time over a longer incubation period and analyzed via HPLC. The expected product of the reaction was found, indicating a catalytic activity of the purified enzymes. When the enzyme YcdW was produced in E. Coli, the formation of glycolic acid was observed in the disrupted cells (Figure 1). The combined evidence shows, that the transformation of E. Coli was successful, leading to the production of functional enzymes. These enzymes then partake in the production pathway of glycolic acid, which was shown in vivo, as well as in vitro.
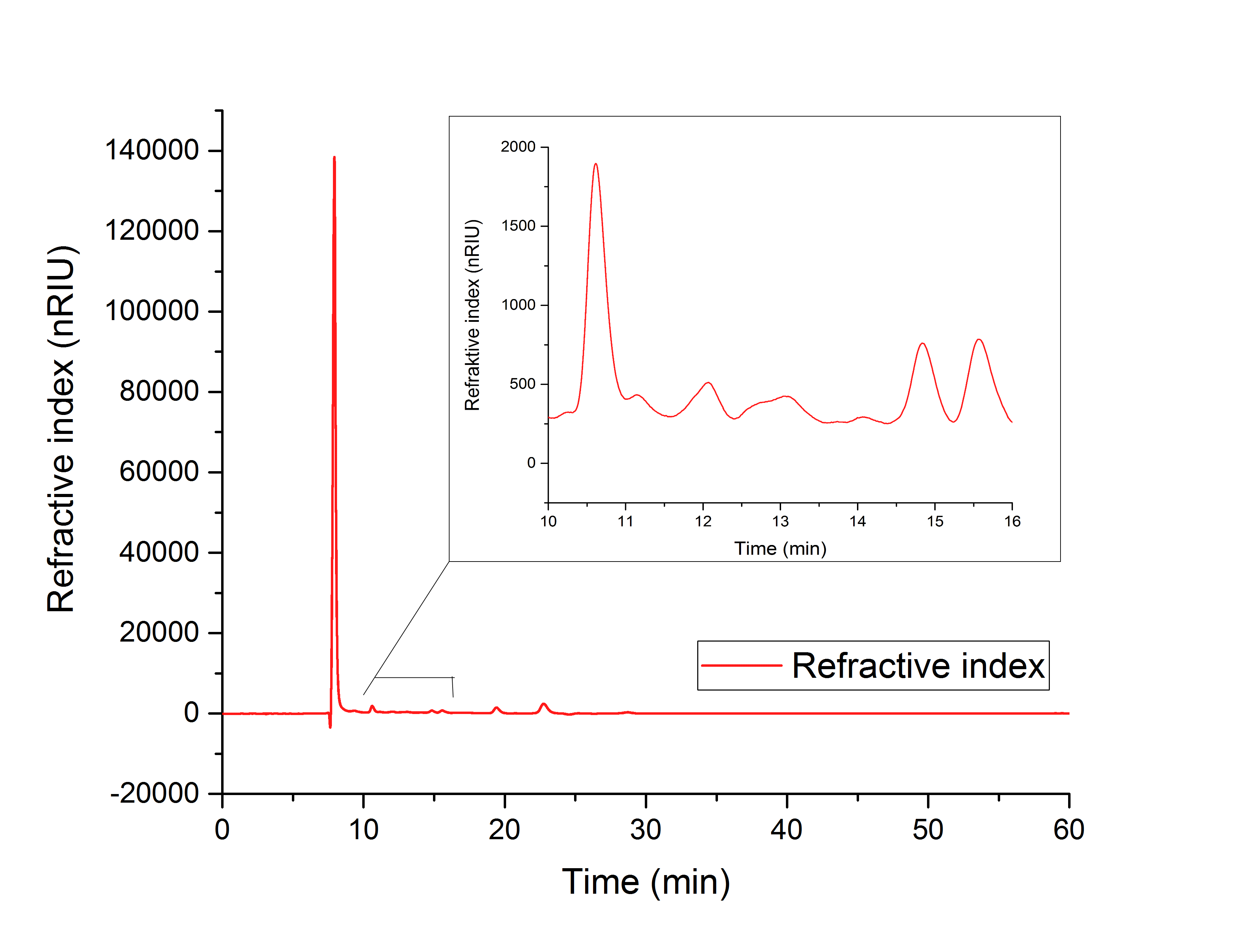
Figure 1: HPLC analysis of supernatant of disrupted ycdW transformed E. coli cells. Glyoxylate and glycolic acid peaks can be seen at 12.066 minutes and 15.568 minutes.
Glycolic acid production in S. cerivisiae
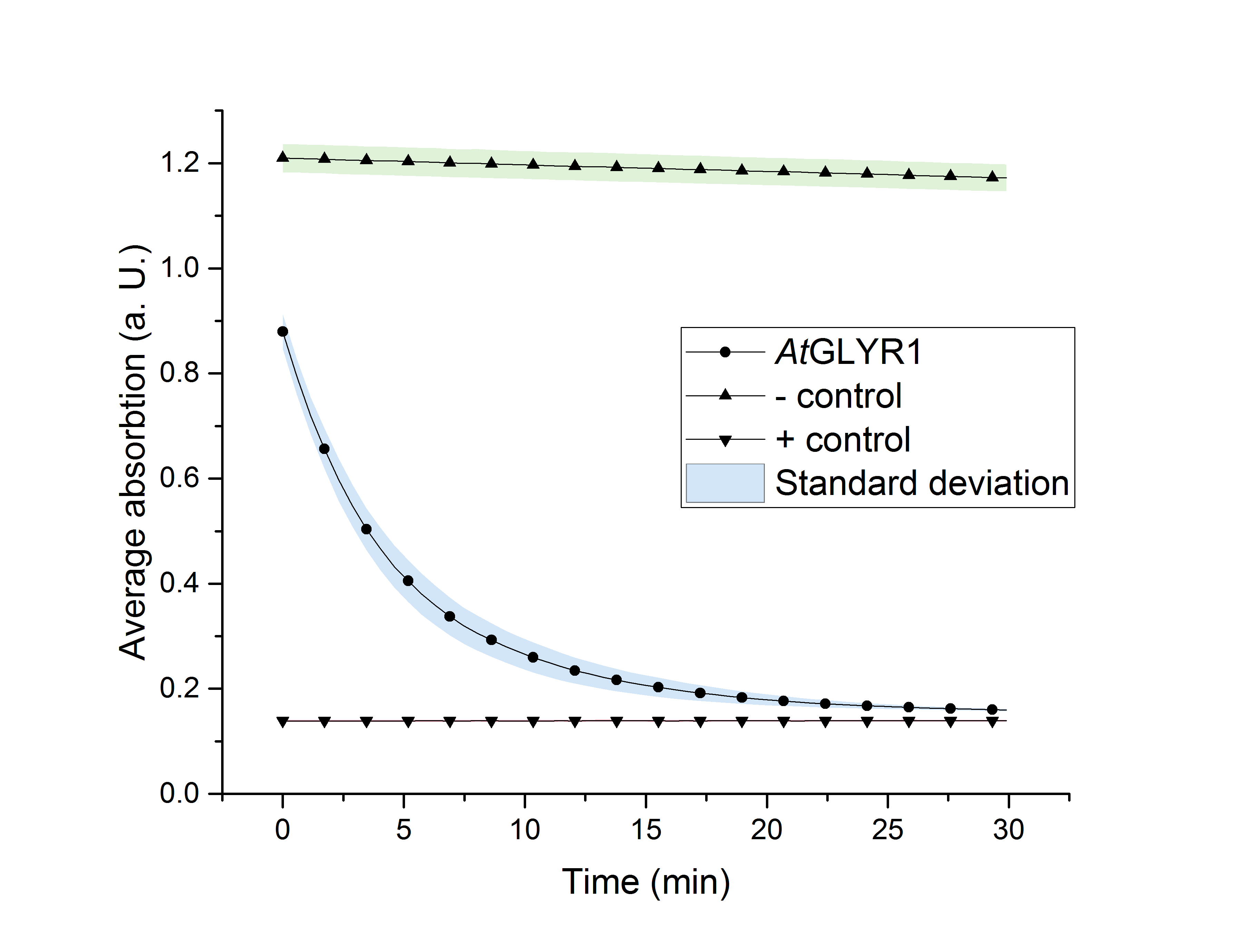
As glycolic acid is one monomer which is needed to produce PLGA, our goal was to produce glycolic acid in S. cerevisiae. It was possible for us to successfully express AtGLYR1 in S. cerevisiae. This was shown by western blot analysis as well as during an enzyme assay. Using a NADPH dependent enzyme assay we could proof the functionality of our purified enzyme (Fig. 2). During the assay AtGLYR1 converts glyoxylate to glycolic acid while turning over NADPH. Therefore, we were able to produce glycolic acid in vitro. Furthermore, we were able to delete several genes from the genome of CEN.PK-1C. This could be helpful to increase the production of glycolic acid in vivo.
Figure 2: NADPH dependent enzyme assay of AtGLYR1. The graph shows the average absorption at 340 nm in correlation to the time (min). The graph shows the turnover of 2,7ng/µl enzyme in the total reaction mix, positive (NADP+) and negative control (NADPH) were included.
ε-caprolactone production in E. coli
The aim of our project was to produce biopolymers, with ε-caprolactone as one potential monomer. To generate this monomer, we successfully overexpressed the genes for heterologous enzymes ADH and CHMO in E. coli. We were able to purify these enzymes using a His-Taq. The purified enzymes were detected via SDS PAGE and western blot. ADH produces NADPH while converting cyclohexanol to cyclohexanone, while CHMO uses NADPH in converting cyclohexanone to ε-caprolactone. An NADPH dependent assay was used, to determine the activity of the enzymes separately, as well as in a combined assay (Figure XX and XX).
Enzyme activity can be shown in both cases (Figure XX).
The equilibrium of the production and depletion of NADPH in the combined assay indicates the synthesis of ε-caprolactone and thus the successful creation of one monomer of our biopolymer.
Manufacturing of polymer
noch zu korrigieren
The fact, that we were able to manufacture polymers like PLGA and PLGC is proved by the GPC analysis. The GPC of PLGC (I) shows a slim distribution of a molecule weight of about 12,000 g/mol, which is, without a doubt, a evidence for polymer.
Und hierhin den Graphen der GPC